· For research use only. Not for human consumption.
For research use only. Not for human consumption.
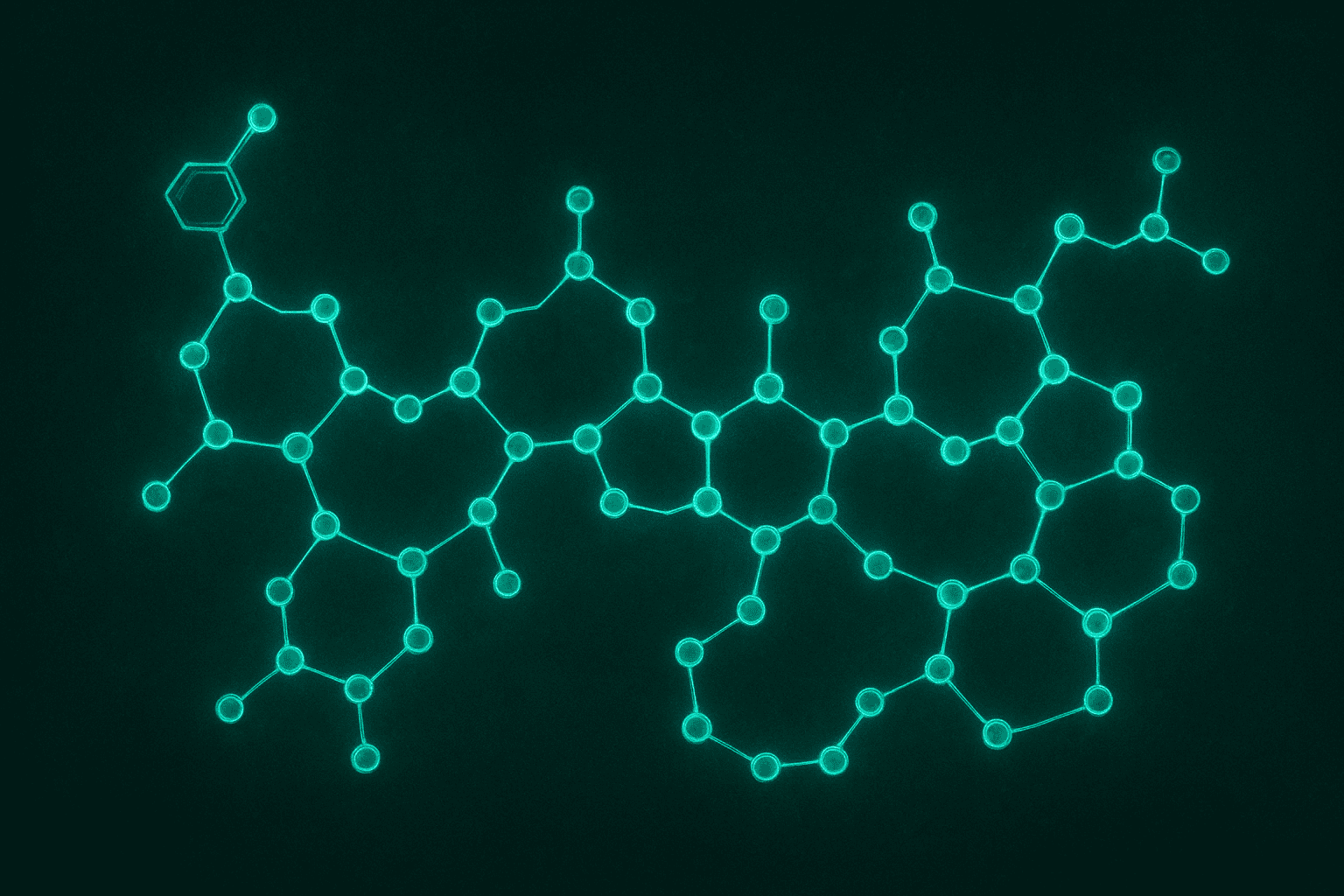
When researchers describe the Tesamorelin amino acid structure, they are talking about a chain of 44 building blocks arranged in a very specific order, with one small chemical modification at the front. That might sound complicated, but the concept is actually simple once you understand what amino acids are and why their arrangement matters. This post will break it all down in plain language.
Tesamorelin’s structure is directly copied from a natural brain signal called GHRH (Growth Hormone Releasing Hormone). The only difference is a protective cap at one end. Understanding this structure helps explain why Tesamorelin behaves the way it does in research settings and why scientists consider it a faithful representation of the natural molecule. You can learn more about our available research peptides in the Alpha Peptides catalog.
Let us start with the fundamentals: what amino acids are and why 44 of them make Tesamorelin a big deal in the peptide world.
TL;DR: Tesamorelin is a 44-amino-acid peptide identical to natural GHRH, plus a trans-3-hexenoic acid cap at the N-terminus. At 44 amino acids, it is large compared to many research peptides (BPC-157 has 15, MOTS-c has 16). Its structure determines its function: the shape allows it to fit the GHRH receptor on pituitary cells. Wang & Tomlinson (2009) described Tesamorelin’s structural design (PMID: 19243281). For research use only. Not for human consumption.
What Are Amino Acids? The Simple Version
Amino acids are the building blocks of every protein and peptide in the body. There are 20 standard amino acids used in human biology, and each one has a unique shape and set of chemical properties. Think of them as 20 different types of Lego bricks. By snapping them together in different orders, the body builds thousands of different molecules, each with a specific job.
When amino acids link together in a chain, the result is called a peptide (for shorter chains) or a protein (for longer ones). The dividing line is roughly 50 amino acids: anything shorter is usually called a peptide, and anything longer is usually called a protein. At 44 amino acids, Tesamorelin falls right near that boundary, making it one of the larger peptides that researchers work with.
The order of amino acids in a chain is critically important. Changing even one amino acid can alter the shape of the entire molecule, which can change how it functions. This is why scientists pay so much attention to the exact sequence when they describe a peptide’s structure.
The Tesamorelin Amino Acid Sequence
Tesamorelin contains all 44 amino acids of natural GHRH in exactly the same order. The sequence starts with tyrosine at position 1 and continues through all 44 positions to the final amino acid. This is not a shortened version or a rearranged version. It is the complete, native GHRH sequence.
What makes it Tesamorelin instead of plain GHRH is one addition: a chemical group called trans-3-hexenoic acid attached to the very first amino acid. This attachment point is called the N-terminus, which is the scientific name for the beginning of a peptide chain (the other end is called the C-terminus).
Wang and Tomlinson (2009) described this structural design in their comprehensive review, noting that Tesamorelin preserves the complete native sequence while adding only the N-terminal modification.
Wang Y, Tomlinson B (2009) reviewed the structural design of Tesamorelin, a human growth hormone releasing factor analogue that retains the full 44-amino-acid GHRH sequence. (PMID: 19243281)
The Trans-3-Hexenoic Acid Modification
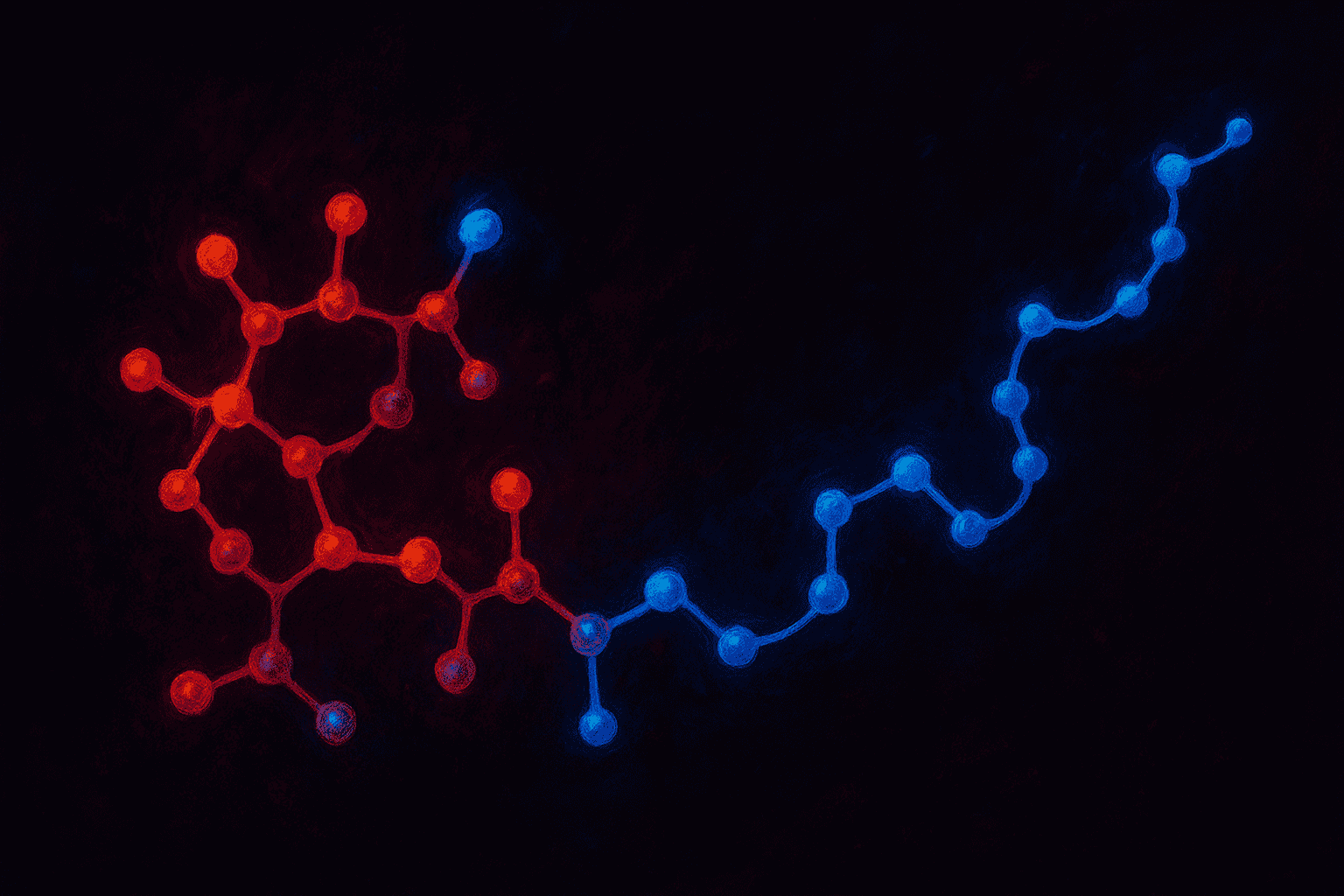
Trans-3-hexenoic acid is a short carbon chain with a double bond. It is a relatively simple molecule in chemistry terms. When it is attached to the N-terminus of the GHRH chain, it acts like a protective cap.
Here is why that matters. The N-terminus is where enzymes called peptidases typically start breaking peptides apart. They grab the beginning of the chain and begin cutting. By placing the trans-3-hexenoic acid group at that spot, scientists created a barrier that peptidases have difficulty grabbing. The result is a molecule that resists breakdown longer than natural GHRH.
Importantly, this cap does not change the way Tesamorelin interacts with the GHRH receptor. The receptor recognizes the amino acid sequence, not the cap. So the molecule still fits into the receptor the same way natural GHRH does. The modification only affects stability, not function.
Why 44 Amino Acids Is Large for a Peptide

In the world of research peptides, 44 amino acids is on the larger side. To put the Tesamorelin amino acid count in perspective, here is how it compares to some other well-known research peptides:
Ipamorelin: 5 amino acids. This growth hormone secretagogue is one of the smallest peptides in active research. Its tiny size makes it very different from GHRH-based peptides in terms of structure and mechanism.
BPC-157: 15 amino acids. This is a commonly referenced research peptide that is roughly one-third the length of Tesamorelin.
MOTS-c: 16 amino acids. Another widely studied peptide that is less than half the length of Tesamorelin.
At 44 amino acids, Tesamorelin is large enough to fold into a complex three-dimensional shape. Smaller peptides tend to be more flexible and floppy, while longer ones have enough amino acid interactions to hold a more defined structure. This structural complexity is part of what allows Tesamorelin to interact specifically with the GHRH receptor and not other receptors in the body.
How Structure Determines Function
One of the most important principles in biology is that the shape of a molecule determines what it can do. This is true for everything from simple vitamins to complex enzymes, and it is absolutely true for peptides like Tesamorelin.
The 44-amino-acid chain of Tesamorelin folds into a specific three-dimensional shape. Parts of the chain form spiral structures called alpha helices. These spirals position key amino acids in exactly the right spots to contact the GHRH receptor on pituitary somatotroph cells. If even a few amino acids were changed or removed, the shape would shift and the molecule might not fit the receptor properly.
This is why Tesamorelin keeps all 44 amino acids intact. While researchers have shown that the first 29 amino acids are sufficient to activate the receptor (which is how Sermorelin works), the full 44-amino-acid chain provides additional stability and maintains the complete native folding pattern. For studies where preserving the exact natural structure is important, Tesamorelin’s full-length design offers an advantage.
Stanley et al. (2011) used Tesamorelin in their research on growth hormone pulsatility, relying on its structural fidelity to the natural GHRH molecule.
Stanley TL et al. (2011) examined the effects of a GHRH analog on endogenous GH pulsatility, leveraging the structural similarity of Tesamorelin to natural GHRH. (PMID: 20943777)
Why Amino Acid Structure Matters for Research

For researchers, knowing the exact amino acid structure of a peptide is not just academic. It has practical implications for every experiment. If the structure is wrong, even by a single amino acid, the results of an experiment could be misleading. This is why reputable peptide suppliers provide detailed Certificates of Analysis that verify the identity and purity of each batch.
Amino acid analysis and mass spectrometry are two common methods used to confirm that a peptide has the correct sequence and the right molecular weight. For a 44-amino-acid peptide like Tesamorelin, these quality checks are especially important because longer chains have more opportunities for errors during the manufacturing process.
Understanding the amino acid structure also helps researchers compare results across studies. When two different laboratories both report using Tesamorelin, they are both referring to the same 44-amino-acid sequence with the same trans-3-hexenoic acid cap. This standardization is essential for building a reliable body of scientific evidence.
Alpha Peptides provides Tesamorelin for qualified researchers, with every batch verified by a third-party Certificate of Analysis (COA) confirming amino acid identity and purity. Visit our research catalog to see all available compounds.
Frequently Asked Questions
How many amino acids does Tesamorelin have?
Tesamorelin has 44 amino acids, the same number as natural GHRH. The sequence is identical to the natural molecule, with the only difference being a trans-3-hexenoic acid group attached to the N-terminus.
Is 44 amino acids considered large for a peptide?
Yes. Most research peptides have far fewer amino acids. BPC-157 has 15, MOTS-c has 16, and Ipamorelin has only 5. At 44 amino acids, Tesamorelin is near the upper boundary of what is typically classified as a peptide rather than a protein.
What is the N-terminus?
The N-terminus is the beginning of a peptide chain. It is the end where enzymes typically start breaking the molecule apart. Tesamorelin has a trans-3-hexenoic acid cap at its N-terminus to slow this breakdown process.
Why does the amino acid sequence matter?
The sequence determines the molecule’s three-dimensional shape, and shape determines function. The correct amino acid sequence allows Tesamorelin to fold properly and fit into the GHRH receptor on pituitary cells. Even small changes to the sequence could alter the shape and disrupt receptor binding.
How is the amino acid sequence of research peptides verified?
Laboratories use methods such as amino acid analysis and mass spectrometry to confirm the identity and purity of peptides. Reputable suppliers provide Certificates of Analysis documenting these results for each batch.
For research use only. Not for human consumption. This material is sold strictly for use in scientific and laboratory research. It is not intended for diagnostic or therapeutic purposes. Alpha Peptides does not endorse or encourage any off-label use.