· For research use only. Not for human consumption.
For research use only. Not for human consumption.
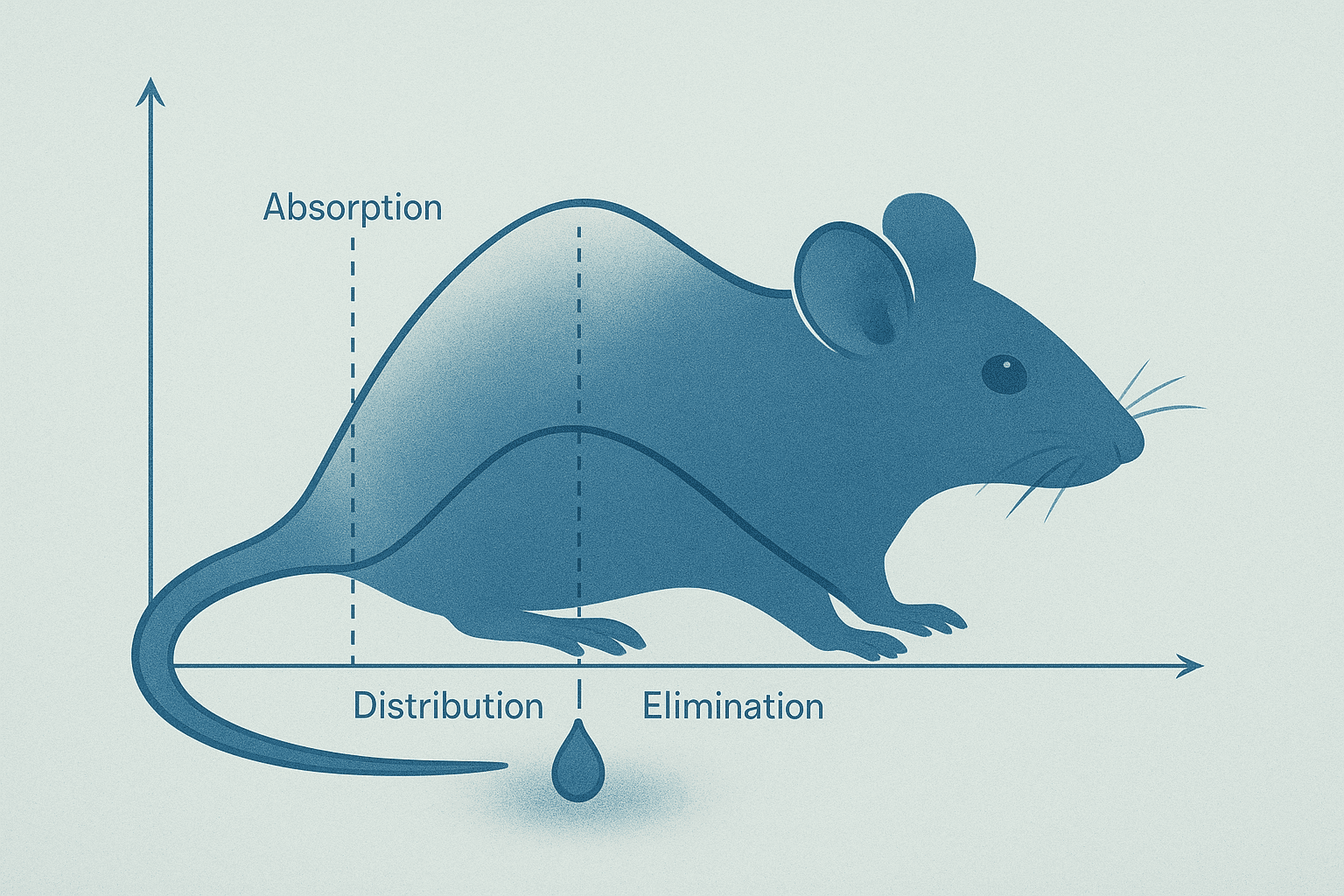
Bioavailability peptide research faces a challenge that most other compound classes do not: peptides get broken down very easily by the body’s digestive enzymes. When researchers talk about bioavailability, they are asking a straightforward question — how much of a compound actually reaches its intended target after it enters a biological system? For peptides, the answer is often “not as much as you would hope,” and understanding why is critical for designing good experiments.
The concept applies to every research peptide in the catalog, from BPC-157’s 15 amino acids to SS-31’s compact 4. Gwyer et al. (2019) reviewed BPC-157 preclinical data across multiple administration routes in animal models (PMID: 30915550). The route of administration — how a compound gets into the system — is one of the biggest factors determining how much of it survives to reach its target.
This guide explains bioavailability in plain language and covers why it matters so much in peptide research specifically. For background on peptide structure and stability, see our peptide bonds guide or our half-life explainer.
TL;DR: Bioavailability measures how much of a compound reaches its target in a biological system. Peptides face unique bioavailability challenges because digestive enzymes (proteases) break them down rapidly. In preclinical research, subcutaneous injection generally provides the highest bioavailability for most peptides. Oral administration presents the biggest challenge. These factors directly influence experimental design and data interpretation. For research use only. Not for human consumption.
What Does Bioavailability Peptide Research Actually Measure?
Bioavailability is the fraction of a compound that makes it to where it needs to go in an active, intact form. If you introduce 100 units of a peptide into a biological system and only 30 units reach the target tissue in working condition, the bioavailability is 30 percent. The other 70 units were lost along the way — broken down, filtered out, or trapped somewhere they could not do anything useful.
For most pharmaceutical compounds, bioavailability is measured relative to intravenous (IV) administration, which delivers 100 percent of a compound directly into the bloodstream. Everything else — oral pills, injections under the skin, nasal sprays — is compared against that IV baseline.
Peptides face a tougher road than most molecules. Their amino acid chains are exactly what digestive enzymes are built to break apart. The body’s proteases (protein-cutting enzymes) evolved specifically to chop peptide bonds. So putting a peptide into a biological system is like sending a paper airplane through a room full of scissors — it has to dodge a lot of threats to arrive in one piece.
Why Do Peptides Have Unique Bioavailability Challenges?
The core problem is this: peptides are made of amino acids, and the body is extremely good at breaking down amino acid chains. Enzymes in the stomach, intestines, blood, and tissues all contain proteases designed to cut peptide bonds. This is normally a good thing — it is how the body digests food proteins and recycles old peptides. But for research purposes, it means peptides are inherently fragile in biological environments.
Three specific challenges stand out:
Enzymatic degradation. Proteases in the gastrointestinal tract, blood, and tissues attack peptide bonds. A peptide that enters the stomach faces pepsin. One that enters the intestines faces trypsin and chymotrypsin. Even peptides that bypass the gut still encounter proteases in the bloodstream. The body treats exogenous peptides the same way it treats food — as something to break down.
Membrane permeability. Most peptides are too large and too hydrophilic (water-loving) to pass through cell membranes easily. Getting from the gut lumen into the bloodstream requires crossing the intestinal epithelium — a barrier that blocks most peptides. Even peptides in the bloodstream may struggle to cross into target tissues.
Rapid clearance. The kidneys filter small molecules out of the blood quickly. Many peptides are small enough to pass through kidney filtration, which means they get excreted before they can accumulate at meaningful concentrations at their target sites.
Routes of Administration and Their Bioavailability Implications
In preclinical research, the route of administration has a dramatic impact on how much of a peptide reaches its target. Different routes expose the peptide to different levels of enzymatic attack and different barriers.
Subcutaneous injection places a peptide just under the skin, where it enters the bloodstream gradually through capillaries. This route avoids the gastrointestinal tract entirely, bypassing the most destructive protease environment. For most research peptides, subcutaneous injection provides relatively high bioavailability compared to other non-IV routes. It is the most commonly used route in preclinical peptide studies.
Intraperitoneal injection delivers a compound into the abdominal cavity, where it is absorbed through the peritoneal membrane. This is common in rodent studies. Bioavailability can be high but variable depending on the peptide’s properties and the specific experimental model.
Intranasal administration delivers a peptide through the nasal mucosa. Some small peptides can cross the nasal epithelium and reach the bloodstream or even the central nervous system through olfactory pathways. Selank and Semax have both been studied using intranasal routes in preclinical models. Kozlovskaya et al. (2003) examined Selank’s effects in animal models and noted intranasal delivery as a method of administration (PMID: 14969422).
Oral administration is the most challenging route for peptides. The stomach’s acidic environment and the intestine’s protease army destroy most peptide chains before they can be absorbed. Oral bioavailability for unmodified peptides is typically very low — often below 1 to 2 percent. This is why oral peptide delivery remains an active area of pharmaceutical research.
Gwyer et al. (2019) reviewed preclinical BPC-157 studies and documented research conducted across multiple routes of administration in animal models, illustrating how administration route affects the peptide’s distribution and the experimental outcomes observed across different tissue types. (PMID: 30915550)
How Bioavailability Connects to Half-Life
Bioavailability and half-life are related but different concepts. Bioavailability asks: “How much of the peptide reaches the target?” Half-life asks: “How long does it stay there before disappearing?” Both factors together determine whether a peptide can accumulate at concentrations high enough to produce measurable effects in an experiment.

A peptide with high bioavailability but a very short half-life reaches the target quickly but disappears fast. A peptide with moderate bioavailability but a longer half-life might accumulate over time and maintain more stable concentrations. Researchers have to consider both variables when designing dosing schedules for preclinical experiments.
Some modifications improve both bioavailability and half-life simultaneously. Adding D-amino acids, PEGylation (attaching polyethylene glycol chains), or cyclization (creating a circular peptide structure) can make a peptide both harder for enzymes to attack and slower to clear from the system. These strategies are commonly used in the design of research-grade peptide analogs.
Why Bioavailability Matters for Experimental Design
For researchers, bioavailability is not an abstract concept — it directly affects practical decisions. How much peptide do you need? Which route of administration fits your experimental model? How do you control for losses during absorption? How do you compare results across studies that used different routes?
A study using subcutaneous injection and a study using oral gavage in rodents may produce different results with the same peptide — not because the peptide itself behaves differently, but because different amounts survived to reach the target tissue. Without accounting for bioavailability differences, comparing data across studies becomes unreliable.

This is one reason why well-designed preclinical studies always report the route of administration, the amount administered, and ideally some measurement of how much compound reached the target tissue. Without that information, the data lacks context. Bioavailability peptide research is not just about the molecule — it is about the entire experimental framework surrounding it.
Alpha Peptides provides research-grade peptides with documented purity and identity, giving researchers a consistent starting material for bioavailability and pharmacokinetic studies. Browse the full research catalog or review third-party testing documentation on our Certificates of Analysis page.
Frequently Asked Questions
Why can’t most peptides be taken orally in research?
The gastrointestinal tract is designed to break down amino acid chains. Stomach acid and intestinal proteases destroy most peptides before they can cross the gut wall into the bloodstream. Oral bioavailability for unmodified peptides is typically below 1 to 2 percent. Researchers working with oral peptide delivery use protective strategies like enteric coatings or chemical modifications, but this remains a major challenge.
Does higher purity improve bioavailability?
Higher purity means a greater percentage of the material in the vial is the intended peptide (rather than synthesis byproducts or contaminants). This gives researchers more accurate control over the amount of active compound they are working with. However, purity does not change the peptide’s inherent susceptibility to enzymatic degradation or membrane barriers — those are properties of the molecule itself.
What does 100 percent bioavailability mean?
It means every molecule of the administered compound reaches the systemic circulation in its active form. Intravenous administration is defined as 100 percent bioavailability because the compound goes directly into the blood. All other routes are compared against this baseline. No non-IV route achieves exactly 100 percent for peptides because some loss always occurs during absorption.
Can peptide modifications improve bioavailability?
Yes. Several strategies have been studied in preclinical research. D-amino acid substitutions make peptides harder for enzymes to recognize and cut. PEGylation attaches large polymer chains that shield the peptide from enzymatic attack and slow kidney clearance. Cyclization creates circular structures that resist protease degradation. These modifications are commonly used in the design of research peptide analogs.
For research use only. Not for human consumption. This article is intended for informational purposes and does not constitute medical advice, dosing guidance, or therapeutic recommendations.