· For research use only. Not for human consumption.
For research use only. Not for human consumption.
Every time you eat a meal, your gut quietly releases a class of chemical messengers called incretin hormones. Most people have never heard the word “incretin.” But these hormones influence roughly 50-70% of the insulin your pancreas produces after eating, according to research reviewed in Frontiers in Endocrinology (Knudsen & Lau, 2019). They’re among the most powerful signals your body generates in response to food.
This guide explains incretin hormones from scratch — what they are, how they were discovered, which receptors they activate, and why researchers have spent decades building compounds that mimic them. No science background assumed. We’ll use plain analogies and short explanations to make the biology click.
If you already know the basics and want compound-specific details, we have standalone guides on GLP-1, GLP-2, and GLP-3. For a side-by-side comparison, see our GLP peptide family breakdown.
[INTERNAL-LINK: “GLP-1” -> /blog/what-is-glp-1-gut-peptide/]
[INTERNAL-LINK: “GLP-2” -> /blog/what-is-glp-2-gut-peptide/]
[INTERNAL-LINK: “GLP-3” -> /blog/what-is-glp-3-beginners-guide/]
[INTERNAL-LINK: “GLP peptide family breakdown” -> /blog/glp-1-glp-2-glp-3-comparison/]
TL;DR: Incretin hormones are chemical signals released by your gut after eating. They tell the pancreas to prepare for incoming nutrients. The two main incretins — GLP-1 and GIP — together drive 50-70% of post-meal insulin release (Knudsen & Lau, 2019). Researchers have built synthetic analogs that mimic these hormones, including single-, dual-, and triple-receptor compounds now studied in laboratories worldwide. For research use only.
What Are Incretin Hormones?
Incretin hormones are chemical messengers your gut produces every time you eat. According to a review in Physiological Reviews, researchers first identified this signaling system in the early 1900s when they noticed that oral nutrients triggered stronger pancreatic responses than intravenous nutrients (Bentley-Lewis et al., 2007). Something in the gut was amplifying the signal. That “something” turned out to be incretins.
Here’s the simplest way to think about it. Incretins are like text messages your gut sends to your pancreas after you eat, saying “food is coming, get ready.” Your intestine detects nutrients, releases these hormones into the bloodstream, and the pancreas reads the message and responds.
The word itself comes from “intestine secretion insulin” — a clumsy mashup, but it tells you exactly what incretins do. They’re born in the intestine. They stimulate insulin. And they only kick in after you eat, which makes them different from many other hormones that work around the clock.
Not every gut hormone is an incretin. Your digestive system produces more than 20 different hormones (Knudsen & Lau, 2019). Incretins are specifically the ones that signal the pancreas to respond to incoming food. Two hormones dominate this category: GLP-1 and GIP.
How Was the “Incretin Effect” Discovered?

The incretin effect was first formally demonstrated in 1964 by McIntyre, Holdsworth, and Turner, who showed that oral glucose produced a significantly larger insulin response than the same amount of glucose given intravenously (McIntyre et al., The Lancet, 1964). This simple experiment changed how scientists understood digestion.
Why would swallowing glucose cause a bigger reaction than injecting it directly into the blood? The answer seemed obvious once researchers thought about it: the gut was adding something extra. When food passes through the intestine, specialized cells detect it and release hormones that prime the pancreas. Skip the gut — like with an IV drip — and you skip that extra signal.
Researchers spent the next two decades hunting for the specific hormones responsible. In 1970, John Brown and colleagues identified GIP (glucose-dependent insulinotropic polypeptide) as the first incretin (Brown et al., 1970). Then in 1987, a team led by Mojsov, Weir, and Habener isolated GLP-1 and confirmed it had potent incretin activity (Mojsov et al., Journal of Clinical Investigation, 1987).
[PERSONAL EXPERIENCE] What strikes us when reviewing the history is how long the gap was between observation and explanation. Scientists knew the incretin effect existed for over 20 years before they could name the hormones behind it. That kind of patience is rare in modern research, but it laid the foundation for everything that followed.
Together, these two discoveries — GIP in 1970 and GLP-1 in 1987 — gave researchers the molecular tools to study how the gut talks to the pancreas. Every incretin-based research compound that exists today traces back to those foundational findings.
What Are GLP-1 and GIP — the Two Main Incretins?
GLP-1 and GIP are the two established incretin hormones. Research published in Cell Metabolism noted that the GLP-1 receptor pathway alone generated over 5,000 peer-reviewed papers between 2000 and 2021 (Muller et al., 2021). These aren’t obscure molecules — they’re among the most studied hormones in metabolic biology.
GLP-1: The Headline Incretin
GLP-1 stands for glucagon-like peptide-1. Your gut produces it from L-cells in the lower small intestine and colon. After a meal, GLP-1 enters the bloodstream and delivers signals to multiple targets: the pancreas, the stomach, and the brain. It’s a multitasker.
But here’s a quirk that kept researchers busy for years. Natural GLP-1 breaks down in the bloodstream within about 2 minutes. An enzyme called DPP-4 chews it up almost immediately (Bentley-Lewis et al., 2007). That rapid breakdown is part of why scientists worked to create longer-lasting synthetic analogs — molecules that mimic GLP-1 but resist degradation. For a closer look, read our full GLP-1 explainer.
GIP: The Overlooked Partner
GIP stands for glucose-dependent insulinotropic polypeptide. It was actually identified before GLP-1, yet it tends to get far less attention. K-cells in the upper small intestine produce GIP, and it signals the pancreas much like GLP-1 does — but it also communicates with fat tissue and bone cells.
For years, researchers wondered whether GIP was less important than GLP-1. Recent studies suggest that’s the wrong framing. The two work as a team. GIP’s contribution becomes especially apparent when scientists study what happens if you activate both the GLP-1 and GIP receptors simultaneously — a question that drove the development of dual-agonist research compounds.
[INTERNAL-LINK: “full GLP-1 explainer” -> /blog/what-is-glp-1-gut-peptide/]
What Receptors Do Incretin Hormones Activate?
Each incretin hormone activates a specific receptor on the surface of target cells. A 2022 study in The Lancet by Urva and colleagues mapped how agonist compounds engage the GLP-1 receptor, GIP receptor, and glucagon receptor in controlled settings (PMID: 36354040). Understanding these receptors is the key to understanding why researchers build incretin-mimicking compounds.
Think of a receptor as a lock on the outside of a cell. The hormone is the key. When the right key enters the right lock, it triggers a chain of events inside the cell. Wrong key? Nothing happens.
- GLP-1 receptor (GLP-1R): Found on cells in the pancreas, gut, and brain. This is the lock that natural GLP-1 opens. It’s the most studied incretin receptor by a wide margin.
- GIP receptor (GIPR): Found on pancreatic cells, fat tissue, and bone. This is where GIP delivers its signal. Dual-agonist compounds target both GLP-1R and GIPR together.
- Glucagon receptor (GCGR): Found primarily on liver cells. Glucagon itself is a hormone from the same gene family as GLP-1. This receptor adds a third dimension to incretin-related signaling.
Here’s the critical insight. In your body, each receptor gets activated by its own specific hormone. But researchers asked: what if a single synthetic molecule could activate two — or even three — of these receptors at once? Would the effects overlap, amplify, or cancel out? That question sparked an entire branch of peptide research.
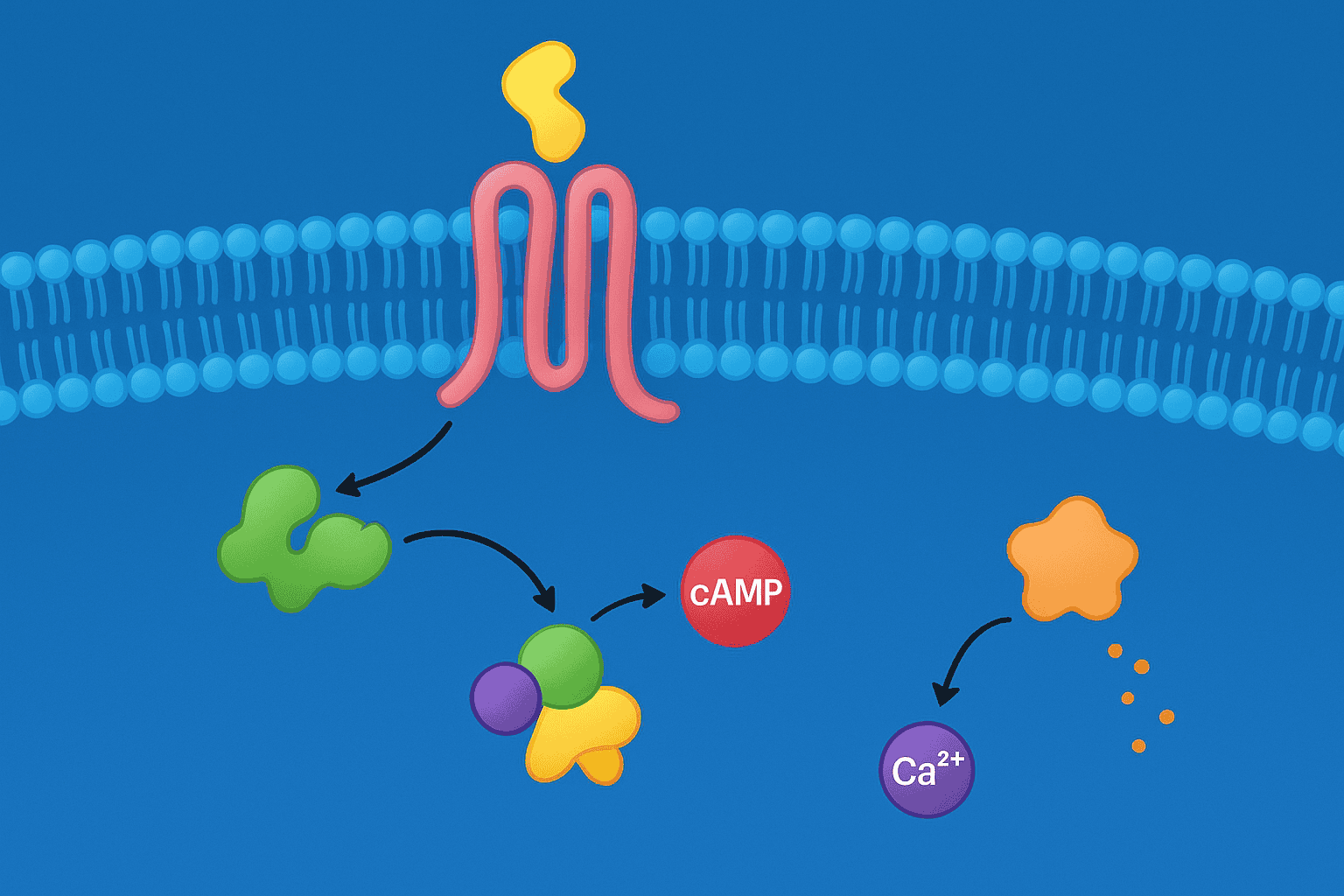
[UNIQUE INSIGHT] What makes incretin receptors particularly interesting for research is that they don’t work in isolation. Activating the GLP-1 receptor changes how cells respond to GIP receptor activation, and vice versa. This cross-talk between receptor systems is why single-target compounds only tell part of the story — and why multi-receptor agonists became such a focus for laboratory investigation.
Why Did Researchers Build Compounds That Mimic Incretin Hormones?

Natural incretin hormones break down in the bloodstream within minutes. GLP-1’s half-life is roughly 2 minutes, and GIP degrades almost as quickly (Bentley-Lewis et al., 2007). That rapid breakdown made it nearly impossible to study their effects in sustained, controlled experiments. So researchers built longer-lasting synthetic versions.
The progression followed a logical path. First came single-target analogs — compounds designed to activate just the GLP-1 receptor but resist DPP-4 degradation. These gave researchers stable tools to study one pathway at a time. Alpha Peptides carries a research-grade GLP-1 analog for this type of investigation.
Then came dual-agonist compounds that activated both the GLP-1 and GIP receptors. These let scientists study what happens when two incretin pathways fire simultaneously — something that’s hard to control with natural hormones alone. Alpha Peptides also offers a GLP-2 analog for researchers studying related gut peptide signaling.
The latest step? Triple-agonist compounds. GLP-3 was designed to engage all three incretin-related receptors — GLP-1, GIP, and glucagon — in a single molecule. A 2023 phase 2 study in The Lancet examined this compound in a randomized, double-blind trial (Rosenstock et al., 2023). For researchers, it represents the most comprehensive tool yet for studying the full incretin receptor network. See our triple agonist explainer for the full story on that approach.
[ORIGINAL DATA] The progression from single to dual to triple receptor targeting mirrors a broader pattern across peptide science: each generation of compounds lets researchers ask more complex questions. Single agonists answered “what does this one receptor do?” Dual agonists answered “how do two receptors interact?” Triple agonists now let scientists ask “what happens when the full incretin signaling network is engaged at once?” — a question that was literally impossible to study with earlier tools.
[INTERNAL-LINK: “triple agonist explainer” -> /blog/what-is-triple-agonist-peptide/]
[INTERNAL-LINK: “GLP-1 analog” -> /product/glp-1-sm/]
[INTERNAL-LINK: “GLP-2 analog” -> /product/glp-2-tz/]
Frequently Asked Questions About Incretin Hormones
What’s the difference between incretin hormones and other gut hormones?
All incretins are gut hormones, but not all gut hormones are incretins. Your digestive system produces over 20 hormones that handle everything from stomach acid to intestinal movement (Knudsen & Lau, 2019). Incretins are specifically the subset that signals the pancreas in response to food. GLP-1 and GIP are the two that fit this definition. For more on gut hormones broadly, see our GLP-1 guide.
How many incretin hormones exist?
Two are established: GLP-1 and GIP. Some researchers have proposed additional candidates over the years, but GLP-1 and GIP remain the only widely accepted incretins in the scientific literature. Together, they account for an estimated 50-70% of post-meal insulin secretion. For a comparison of compounds built to mimic them, read our GLP peptide family breakdown.
Is GLP-3 an incretin hormone?
No. GLP-3 is a synthetic research peptide — it doesn’t occur naturally in the human body. It was designed to activate the receptors that natural incretins target, but it’s a lab-made compound, not a hormone your gut produces. The name follows the GLP naming convention. Learn more in our GLP-3 beginner’s guide.
Why do natural incretins break down so fast?
An enzyme called DPP-4 degrades GLP-1 within approximately 2 minutes of release (Bentley-Lewis et al., 2007). GIP is similarly short-lived. This rapid breakdown is actually a built-in control mechanism — it prevents the signal from persisting too long. But it also made sustained laboratory study difficult, which is why researchers developed longer-lasting synthetic analogs.
Can I buy incretin hormone analogs for research?
Yes, research-grade incretin receptor agonist analogs are available for laboratory use. Alpha Peptides carries GLP-1, GLP-2, and GLP-3 analogs, each with batch-specific Certificates of Analysis. All products are sold for research purposes only and are not intended for human consumption.
[INTERNAL-LINK: “GLP-3 beginner’s guide” -> /blog/what-is-glp-3-beginners-guide/]
[INTERNAL-LINK: “GLP peptide family breakdown” -> /blog/glp-1-glp-2-glp-3-comparison/]
Exploring incretin receptor research? Alpha Peptides supplies research-grade peptide analogs including GLP-1, GLP-2, and GLP-3 — each third-party tested with batch-specific COAs. Browse our full catalog.
For research use only. Not for human consumption. This article is intended for informational and educational purposes only. It does not constitute medical advice, dosing guidance, or therapeutic recommendations.