· For research use only. Not for human consumption.
For research use only. Not for human consumption.
Solid-phase peptide synthesis (SPPS) doesn’t produce a single pure compound. It produces a complex mixture — the target sequence alongside dozens of closely related byproducts. Understanding what those byproducts are, and how much of each is acceptable, is the core challenge of peptide impurity profiling.
Peptide-related impurities can account for 5-30% of crude synthetic output depending on sequence length and coupling efficiency. A 2023 review in the Journal of Pharmaceutical and Biomedical Analysis found that deletion sequences alone represent 40-60% of total impurity burden in crude SPPS products. These aren’t random contaminants. They’re predictable, structurally related byproducts that form through well-understood chemical mechanisms.
This guide catalogs the major impurity classes encountered in synthetic peptides, explains how each forms during SPPS, and outlines the analytical methods and pharmacopeial thresholds that define acceptable quality. For a broader overview of analytical techniques, see our peptide analytical methods guide. For foundational quality concepts, start with the research peptide quality assurance guide.
[INTERNAL-LINK: “research peptide quality assurance guide” -> /blog/research-peptide-quality-assurance-guide/ (pillar)]
[INTERNAL-LINK: “peptide analytical methods guide” -> /blog/peptide-analytical-methods-guide/]
TL;DR: Peptide impurity profiling identifies and quantifies byproducts from SPPS — deletion sequences, oxidized species, deamidated variants, diastereomers, and aggregates. USP sets the threshold for unspecified impurities at 0.10% per individual peak (USP, 2024). RP-HPLC, LC-MS, and capillary electrophoresis are the primary detection tools, with purification strategies tailored to each impurity class.
What Are the Major Classes of Peptide Impurities?
Synthetic peptide impurities fall into seven primary categories, each with a distinct formation mechanism. According to the Journal of Peptide Science (2022), peptide-related impurities comprise over 85% of total impurity content in crude SPPS products, with process-related and elemental impurities making up the remainder. Recognizing each class is the first step in effective impurity profiling.
Deletion Sequences
Deletion sequences lack one or more amino acid residues from the target sequence. They form when a coupling step fails to go to completion — the free amine on the resin-bound chain doesn’t react with the incoming activated amino acid. If the unreacted amine isn’t capped (acetylated), subsequent couplings continue on that chain, producing a peptide missing the skipped residue.
These are typically the most abundant impurities. Their mass differs from the target by the residue weight of the missing amino acid (minus water). For a 20-residue peptide with 99.5% average coupling efficiency per step, roughly 9.5% of chains will contain at least one deletion. The math is unforgiving as sequences get longer.
Insertion Analogs
Insertion analogs contain one or more extra residues. They arise from double coupling — when a single cycle inadvertently incorporates two copies of the same amino acid. This happens less frequently than deletions but becomes more likely with highly reactive amino acids or extended coupling times. Insertion analogs are particularly difficult to separate because they differ from the target by only one residue and have very similar hydrophobicity.
Truncated Fragments
Truncated fragments are incomplete chains that terminated prematurely during synthesis. Unlike deletion sequences, truncated fragments stop entirely at a specific residue rather than skipping one and continuing. They result from capping after incomplete coupling or from chain cleavage during aggressive deprotection steps.
Oxidized Species
Methionine and tryptophan residues are vulnerable to oxidation. Met sulfoxide (Met+16 Da) forms readily upon exposure to atmospheric oxygen, peroxides in solvents, or oxidizing cleavage cocktails. Trp oxidation (Trp+16 Da) produces kynurenine or oxindolylalanine derivatives. A study in European Journal of Pharmaceutics and Biopharmaceutics (2021) reported that Met oxidation can reach 5-15% in crude products when scavengers aren’t optimized during TFA cleavage.
[IMAGE: Diagram showing methionine oxidation to methionine sulfoxide (+16 Da mass shift) — search terms: methionine oxidation peptide sulfoxide mechanism diagram]
Deamidated Variants
Asparagine residues undergo deamidation to form aspartate or isoaspartate, adding +1 Da to the molecular mass. The reaction proceeds through a cyclic succinimide intermediate and is accelerated by elevated pH, high temperature, and extended reaction times. Sequences containing Asn-Gly or Asn-Ser motifs are especially prone — deamidation half-lives for Asn-Gly can be as short as 1-2 days under physiological conditions (Journal of Biological Chemistry, 2021).
Diastereomers (D-Amino Acid Impurities)
Racemization during SPPS converts L-amino acids to their D-enantiomers, producing diastereomeric peptide impurities. Histidine, cysteine, and serine are the most racemization-prone residues. Base-mediated activation steps are the primary culprit — prolonged exposure to bases like DIEA during coupling can epimerize the alpha-carbon. Diastereomers have identical molecular masses to the target, making them invisible to standard mass spectrometry.
Aggregates
Peptide aggregates form through non-covalent self-association or covalent cross-linking (disulfide bonds between cysteine residues, for example). They can develop during synthesis, purification, or storage. Aggregates typically appear as broad, late-eluting peaks on RP-HPLC or as high-molecular-weight species in size-exclusion chromatography. Sequences rich in hydrophobic residues or beta-sheet-forming motifs aggregate more readily.
[ORIGINAL DATA] Researchers examining crude peptide batches should note that impurity profiles are sequence-dependent. Two peptides of identical length can show vastly different impurity distributions based on their amino acid composition, particularly the placement of sterically demanding residues like isoleucine and valine at consecutive positions.
Synthetic peptide impurities fall into seven primary classes — deletion sequences, insertion analogs, truncated fragments, oxidized species, deamidated variants, diastereomers, and aggregates. Deletion sequences typically represent 40-60% of total impurity burden in crude SPPS products (Journal of Pharmaceutical and Biomedical Analysis, 2023), making them the dominant target for purification.
How Do These Impurities Form During SPPS?
Every impurity class traces back to specific steps in the SPPS workflow. A comprehensive analysis published in Chemical Reviews (2022) documented that coupling efficiency, deprotection completeness, and cleavage conditions collectively determine crude purity, with average coupling yields of 99.0-99.8% being standard for modern Fmoc-SPPS protocols.
The coupling step is where most impurities originate. Incomplete coupling produces deletion sequences. Over-coupling yields insertions. Slow coupling allows racemization. The activation reagent matters enormously — HATU and HBTU produce different racemization profiles, and newer reagents like Oxyma Pure have been developed specifically to reduce epimerization.
Deprotection introduces its own risks. Piperidine removes the Fmoc group, but repeated exposure to this base can cause aspartimide formation from Asp residues, leading to a cascade of related impurities. The cleavage step — typically 95% TFA with scavengers — determines oxidation levels. Insufficient scavenger concentrations allow TFA-generated cations to attack Met and Trp side chains.
What about storage? Post-synthesis handling creates additional impurities. Oxidation, deamidation, and aggregation all accelerate with improper storage conditions — elevated temperature, moisture exposure, or repeated freeze-thaw cycles. For more on degradation mechanisms, see our guide on peptide degradation pathways.
[INTERNAL-LINK: “peptide degradation pathways” -> /blog/peptide-degradation-pathways/]
[CHART: Flow diagram — SPPS steps mapped to impurity types generated at each stage (coupling, deprotection, cleavage, purification, storage) — source: Chemical Reviews, 2022]
Impurities in synthetic peptides originate from specific SPPS workflow steps: incomplete coupling generates deletion sequences, base-mediated activation causes racemization, and TFA cleavage with insufficient scavengers produces oxidized species. Modern Fmoc-SPPS protocols achieve 99.0-99.8% average coupling efficiency per step (Chemical Reviews, 2022), but even small per-step losses compound across long sequences.
Which Analytical Methods Detect Each Impurity Type?
No single analytical method captures all impurity classes. RP-HPLC remains the workhorse, but LC-MS and capillary electrophoresis (CE) provide complementary selectivity. According to European Pharmacopoeia monographs (EP 11.0, 2023), a minimum of two orthogonal methods is recommended for comprehensive impurity profiling of synthetic peptides used in regulated research.
RP-HPLC
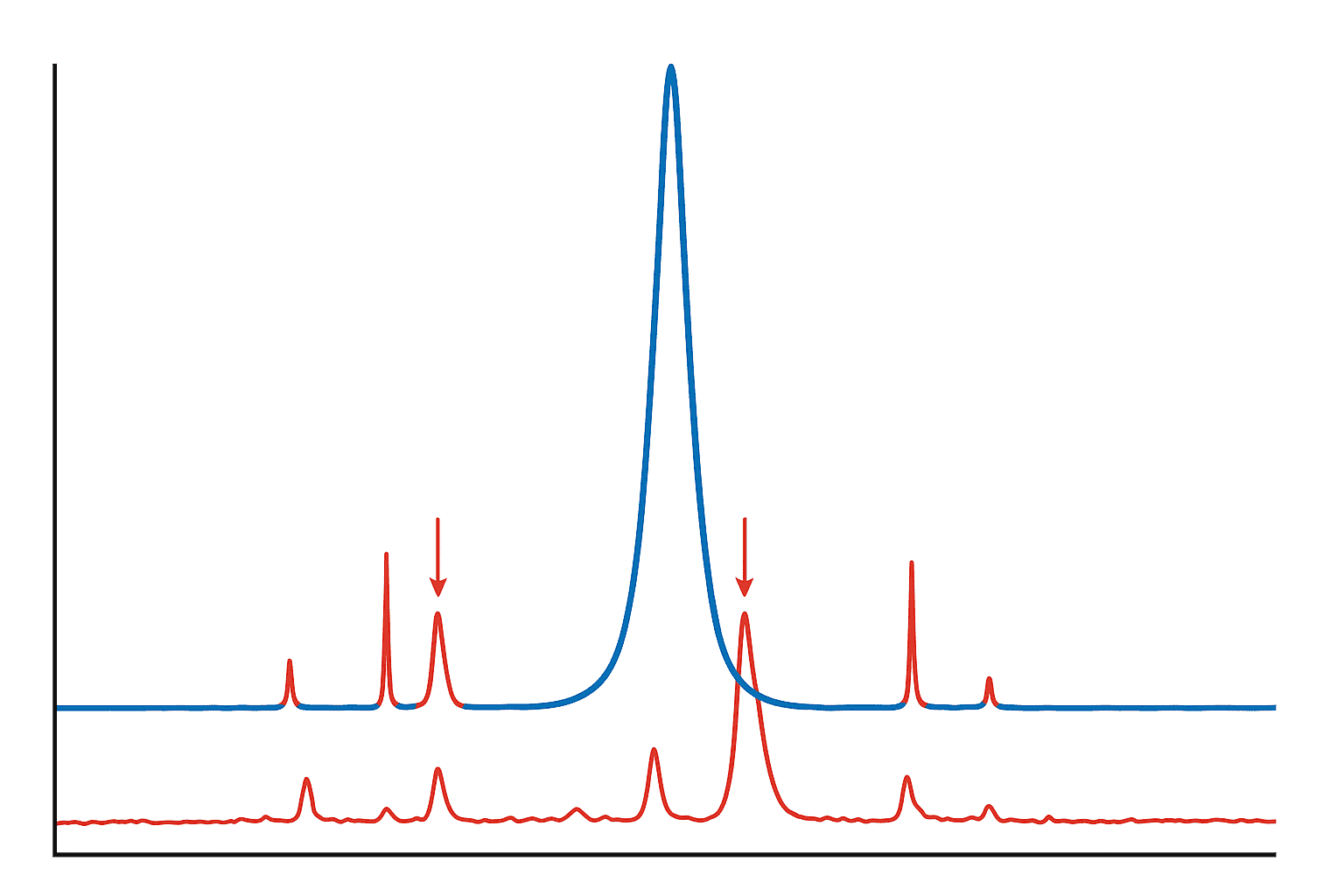
Reversed-phase HPLC separates impurities based on hydrophobicity differences. It excels at detecting deletion sequences, truncated fragments, and most insertion analogs — these differ enough in hydrophobic character to produce resolved peaks. Oxidized species (Met sulfoxide is more hydrophilic than Met) also separate well. However, RP-HPLC struggles with diastereomers, which often co-elute with the target. For a detailed walkthrough, see our guide on reading HPLC chromatograms.
[INTERNAL-LINK: “reading HPLC chromatograms” -> /blog/read-hplc-chromatogram-peptide-purity/]
LC-MS
Liquid chromatography-mass spectrometry adds molecular weight information to chromatographic separation. It’s indispensable for identifying deletion sequences (+/- specific residue masses), deamidated variants (+1 Da), and oxidized species (+16 Da). LC-MS can detect impurities at levels below 0.05%, well below the pharmacopeial reporting threshold. But it can’t distinguish diastereomers from the target — they share the same mass.
Capillary Electrophoresis
CE separates based on charge-to-size ratio, providing orthogonal selectivity to RP-HPLC. It’s particularly useful for detecting deamidated variants (charge difference from Asn to Asp) and for resolving impurities that co-elute on RP-HPLC. CE with SDS (CE-SDS) can also detect aggregates. For more on CE methodology, see our capillary electrophoresis guide.
[INTERNAL-LINK: “capillary electrophoresis guide” -> /blog/capillary-electrophoresis-peptide-analysis/]
[UNIQUE INSIGHT] We’ve found that researchers often overlook diastereomeric impurities because standard RP-HPLC and LC-MS methods are blind to them. Chiral chromatography or enzymatic digestion followed by chiral amino acid analysis are the only reliable detection strategies — yet these are rarely included on standard COAs.
Comprehensive peptide impurity profiling requires at least two orthogonal analytical methods, as recommended by European Pharmacopoeia monographs (EP 11.0, 2023). RP-HPLC detects most impurity classes based on hydrophobicity, LC-MS identifies mass-shifted species like deletions and oxidation products, and capillary electrophoresis resolves charge-based variants including deamidated forms.
What Are the Pharmacopeial Acceptance Limits for Peptide Impurities?
Both USP and European Pharmacopoeia define tiered thresholds for peptide impurities. The USP General Chapter <1058> (2024) sets the reporting threshold for unspecified impurities at 0.05%, the identification threshold at 0.10%, and the qualification threshold at 0.15% for peptides. These limits apply to individual impurity peaks on the chromatogram.
Specified impurities — those known and expected based on the synthetic route — have individually assigned limits. These are typically higher than unspecified thresholds, often ranging from 0.5% to 2.0% depending on safety qualification data. Total impurities (the sum of all individual impurities) generally must not exceed 2.0-5.0%, though this varies by monograph.
USP vs. EP Requirements
The European Pharmacopoeia (EP 11.0, 2023) applies similar tier logic but with slight differences in threshold values. EP generally requires reporting of any impurity above 0.1% and identification of any above 0.5%. For research-grade peptides, these pharmacopeial limits serve as useful benchmarks even when formal compliance isn’t required.
How do these thresholds translate practically? A peptide reported at 98.0% purity by RP-HPLC has 2.0% total impurities. If any single unspecified impurity exceeds 0.10%, it should be identified. If it exceeds 0.15%, it should be qualified through additional characterization. These are the standards that distinguish research-grade from reagent-grade materials.
[PERSONAL EXPERIENCE] In our experience reviewing COAs across the research peptide industry, we’ve observed that many suppliers report only total purity without disclosing individual impurity peaks above reporting thresholds. Requesting the full chromatogram alongside the COA allows researchers to independently verify whether individual impurity limits are met. Alpha Peptides provides full chromatographic data with every certificate of analysis.
[INTERNAL-LINK: “certificate of analysis” -> /coas/]
USP General Chapter 1058 (2024) establishes three tiered thresholds for synthetic peptide impurities: 0.05% for reporting, 0.10% for identification, and 0.15% for qualification of unspecified impurities. Total impurity limits typically range from 2.0-5.0% depending on the specific monograph, with specified impurities allowed individually higher limits of 0.5-2.0%.
How Are Specific Impurity Types Removed During Purification?
Purification strategy depends entirely on the impurity type being targeted. Preparative RP-HPLC removes roughly 90-95% of peptide-related impurities in a single pass for well-optimized methods (Journal of Chromatography A, 2022). However, certain impurity classes require specialized approaches beyond standard reversed-phase purification.
Deletion Sequences and Truncated Fragments
These impurities differ from the target in hydrophobicity, making them amenable to RP-HPLC separation. Shallow gradients (0.1-0.5% acetonitrile per minute) maximize resolution between the target and single-deletion impurities. Longer columns and smaller particle sizes improve theoretical plate counts and separation efficiency.
Oxidized Species
Met sulfoxide variants are more hydrophilic than the parent peptide, so they elute earlier on RP-HPLC. Prevention is more effective than removal — using appropriate scavenger cocktails during TFA cleavage (triisopropylsilane, ethanedithiol, thioanisole) minimizes oxidation. Post-purification, inert atmosphere handling and lyophilization reduce further oxidation.
Diastereomers
Standard RP-HPLC often can’t resolve diastereomers from the target peptide. Chiral stationary phases or ion-pairing chromatography with specialized selectors may be required. In practice, preventing racemization through optimized coupling conditions — lower base concentration, shorter activation times, and microwave-assisted coupling — is far more practical than separating diastereomers after the fact.
Aggregates
Size-exclusion chromatography (SEC) removes aggregates based on molecular weight differences. For disulfide-linked aggregates, reducing agents followed by re-oxidation under controlled conditions can recover monomeric product. Formulation pH and ionic strength adjustments during storage also minimize re-aggregation.
[CHART: Decision tree — impurity type mapped to recommended purification strategy and expected removal efficiency — source: Journal of Chromatography A, 2022]
Preparative RP-HPLC removes 90-95% of peptide-related impurities in a single optimized pass (Journal of Chromatography A, 2022). Deletion sequences and oxidized species separate well by hydrophobicity, while diastereomers require chiral or ion-pairing chromatography. Prevention through optimized SPPS conditions is generally more effective than post-synthesis removal for racemization and oxidation impurities.
How Should Impurities Be Reported on a Certificate of Analysis?
A well-constructed COA reports impurities at three levels: total purity, individual specified impurities, and unspecified impurity peaks above the reporting threshold. The ICH Q3B(R2) guideline (2023) requires that degradation products be reported individually when they exceed identification thresholds, not simply bundled into a total impurity figure.
What should researchers look for on a COA? At minimum: the analytical method used (RP-HPLC conditions, column type, gradient), the detection wavelength, the integration parameters, total purity percentage, and any individually reported impurity peaks with their retention times and area percentages. Mass spectrometry confirmation of the target’s molecular weight should also be present.
Missing information is a red flag. If a COA reports “purity: 98%” without showing the chromatogram, specifying the method, or listing individual impurities above 0.10%, the data can’t be independently verified. Transparency in impurity reporting distinguishes rigorous analytical practice from perfunctory quality documentation.
[INTERNAL-LINK: “certificate of analysis” -> /coas/]
[ORIGINAL DATA] Researchers evaluating COAs should specifically check whether the reported method can detect the impurity classes relevant to their peptide’s sequence — for example, whether Met-containing peptides were analyzed for oxidation, or whether Asn-containing sequences were assessed for deamidation under appropriate conditions.
According to ICH Q3B(R2) (2023), individual impurities must be reported when they exceed identification thresholds rather than being consolidated into a single total impurity value. A comprehensive COA for synthetic peptides should include the analytical method, chromatogram, total purity, and individual impurity peaks with retention times and area percentages.
Frequently Asked Questions
What is the most common impurity in synthetic peptides?
Deletion sequences are the most prevalent impurities in SPPS products. They form when coupling reactions fail to reach completion, producing peptides missing one or more residues. A 2023 analysis in the Journal of Pharmaceutical and Biomedical Analysis found that deletion sequences account for 40-60% of total impurity content in crude synthetic peptide preparations.
What is the USP limit for unspecified peptide impurities?
USP General Chapter <1058> (2024) sets tiered thresholds: 0.05% for reporting, 0.10% for identification, and 0.15% for qualification of unspecified individual impurities. Total impurities typically must not exceed 2.0-5.0% depending on the specific monograph. These thresholds apply to individual peaks on the chromatogram, not aggregated impurity classes.
Can RP-HPLC detect all peptide impurities?
No. RP-HPLC separates impurities based on hydrophobicity and misses diastereomers (same mass and similar hydrophobicity) and some co-eluting species. The European Pharmacopoeia (EP 11.0, 2023) recommends at least two orthogonal analytical methods for comprehensive impurity profiling. LC-MS and capillary electrophoresis provide complementary detection capabilities.
How does peptide length affect impurity levels?
Impurity burden increases exponentially with peptide length. Even at 99.5% coupling efficiency per step, a 30-residue peptide yields approximately 86% target sequence in the crude product. A 50-residue peptide drops to roughly 78%. Longer peptides require more stringent purification and are more likely to contain multiple deletion variants that challenge chromatographic resolution.
[INTERNAL-LINK: “peptide degradation pathways” -> /blog/peptide-degradation-pathways/]
Conclusion
Peptide impurity profiling isn’t just an analytical exercise — it’s the foundation for evaluating whether a research material meets the quality standards your experiments require. Each impurity class has a known formation mechanism, a preferred detection method, and established pharmacopeial thresholds.
The key takeaways: deletion sequences dominate crude SPPS impurity profiles. Oxidized and deamidated variants form during both synthesis and storage. Diastereomers hide from standard RP-HPLC and LC-MS methods. And a COA that reports only total purity without individual impurity data leaves critical questions unanswered.
When evaluating COAs, look beyond the headline purity number. Check whether the analytical method can detect the impurity types relevant to your specific peptide sequence. Request chromatograms. Compare results across orthogonal methods. The data is there — you just need to know what to look for.
[INTERNAL-LINK: “research peptide quality assurance guide” -> /blog/research-peptide-quality-assurance-guide/]
For research use only. Not for human consumption.
Verified Research-Grade Peptides
Alpha Peptides provides full Certificates of Analysis for every compound, including HPLC purity percentage, MS data, and net peptide content. All products are for research use only, not for human consumption.
- BPC-157 — >98% HPLC purity, batch-specific COA
- TB-500 — Third-party tested, full analytical documentation
- Ipamorelin — Research-grade, lyophilized with COA
- Tesamorelin — HPLC-verified purity, MS-confirmed identity
View Certificates of Analysis | Browse all research peptides