· For research use only. Not for human consumption.
For research use only. Not for human consumption.
You’ve got a GLP-3 COA in front of you — a sheet full of numbers, abbreviations, and lab jargon. It looks like it was written for scientists. That’s because it was. But here’s the thing: you don’t need a science degree to understand what it’s telling you.
A Certificate of Analysis is basically a report card for your peptide. It tells you what’s inside the vial, how pure it is, and whether it’s actually the compound you ordered. Once you know what each section means, reading a GLP-3 COA takes about two minutes.
This guide walks you through every section of a typical COA in plain, everyday language. No chemistry background required. If you’re new to GLP-3 entirely, our beginner’s guide to GLP-3 is a good starting point. For a broader overview of COAs across all peptides, see our general COA explainer.
TL;DR: A GLP-3 COA shows you five critical pieces of information: HPLC purity (look for 98%+), mass spectrometry identity confirmation, batch number, lab name, and testing date. According to ISO/IEC 17025 standards, third-party lab testing provides the most reliable verification. Always match the batch number on the COA to the label on your vial. For research use only.
What Is a GLP-3 COA, Exactly?
A GLP-3 COA is a document produced by a testing laboratory that verifies the contents of a specific batch of GLP-3 peptide. The U.S. Pharmacopeia defines a COA as a report demonstrating a product meets its stated specifications (USP General Chapter 1080, 2023). In plain terms, it’s proof that what’s in the vial matches what’s on the label.
Think of buying a used car. You can look at the paint and kick the tires, but you don’t really know the car’s condition until you see the inspection report. A COA is that inspection report — except for peptides.
GLP-3 is a triple incretin receptor agonist analog, which makes accurate identification especially important. Its molecular structure is complex, and without proper testing documentation, there’s no visual way to confirm what’s in a vial of white lyophilized powder.
What Does the Purity Percentage Tell You?

The purity number is usually the first thing people look for on a GLP-3 COA — and for good reason. According to AOAC International’s validation guidelines, HPLC (high-performance liquid chromatography) delivers reproducibility above 99% under controlled conditions (AOAC International, 2022). That makes it the industry gold standard for measuring how clean a sample is.
So what does “98.5% purity” actually mean? It means that 98.5 parts out of every 100 in your sample are the target peptide. The remaining 1.5% is everything else — leftover solvents, synthesis byproducts, or trace impurities.
Here’s a simple analogy. Imagine a jar of 100 jellybeans. If 98 are the flavor you ordered and 2 are random rejects, that’s 98% purity. For research-grade GLP-3, you want that number at 98% or higher. Anything below 95% means enough unknown material is present to potentially interfere with experimental results.
The purity is determined by HPLC testing — a lab technique that separates and measures each component in the sample. Want to understand exactly how HPLC works? We’ve got a dedicated breakdown of HPLC testing.
How Does Mass Spectrometry Confirm Identity?

Purity tells you how clean the sample is. Mass spectrometry tells you what the sample actually is. These are two separate questions. A 2006 review in Analytical Chemistry described mass spectrometry as the definitive tool for molecular identity confirmation, with accuracy within 0.01 daltons on modern instruments (Domon and Aebersold, 2006).
Every peptide has a unique molecular weight — a kind of fingerprint determined by its amino acid sequence. Mass spec measures the weight of the molecules in your vial and compares it to the expected number. If they match, you’ve confirmed it’s actually GLP-3.
Why does this matter? Because HPLC alone can’t confirm identity. A vial could be 99% pure — but 99% pure of the wrong compound. Mass spectrometry closes that gap. If your GLP-3 COA only shows HPLC data without mass spec confirmation, it’s essentially half a document.
Think of it like a passport check at the airport. HPLC confirms you’re wearing clean clothes (purity). Mass spec confirms you’re actually the person whose name is on the ticket (identity). You need both.
What Do Batch Numbers, Lab Names, and Dates Tell You?

Three details on a GLP-3 COA often get overlooked, but they’re critical for verification. ISO/IEC 17025 — the international standard for testing labs — requires that accredited laboratories document every test with traceable batch identifiers and dates (ISO/IEC 17025, 2017).
Batch or Lot Number
This number ties the COA to one specific production run. The batch number on your COA should match the batch number on your vial’s label. If they don’t match — or if there’s no batch number at all — the document could apply to a completely different product. No match, no proof.
Testing Laboratory Name
A legitimate COA names the lab that performed the testing. You should be able to search that lab online and verify it exists. Better yet, check whether the lab holds ISO/IEC 17025 accreditation — that means it’s been independently audited for competence and impartiality.
Date of Analysis
The testing date tells you when the analysis was performed. A COA that’s years old may not reflect the current condition of the product, especially if the peptide has been stored for a long time. Recent testing dates — ideally within a few months of your purchase — are a good sign.
How Can You Spot a Fake or Generic COA?
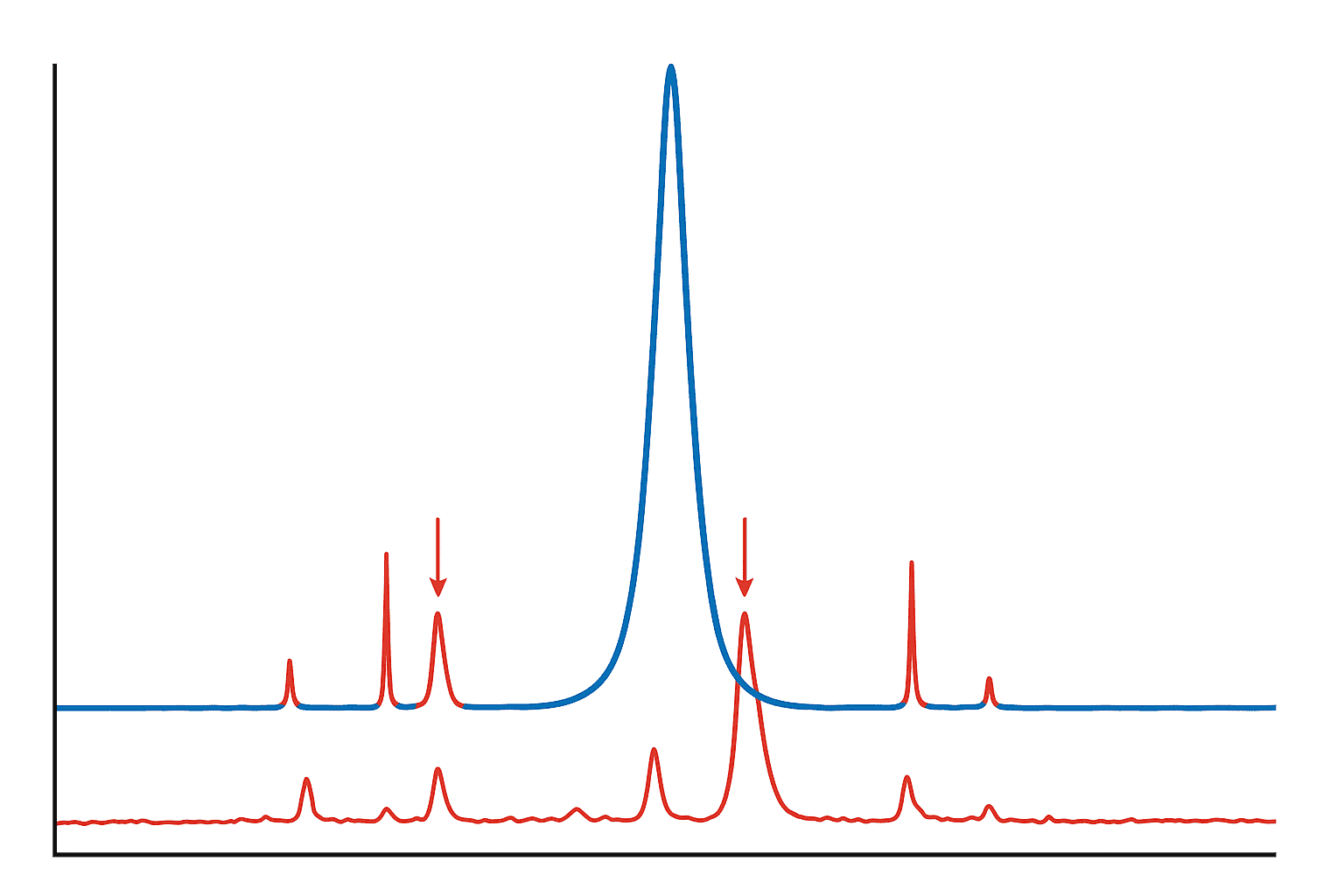
Unfortunately, not every COA floating around online is legitimate. A 2020 analysis by the Partnership for Safe Medicines found that roughly 1 in 5 online research chemical suppliers failed to provide verifiable testing documentation when requested (Partnership for Safe Medicines, 2020). Here’s what to watch for.
Red Flags That Should Make You Pause
- No batch number. A COA without a batch number is generic — it doesn’t correspond to any specific production lot. It’s essentially a template.
- No lab name or an unverifiable lab. If the lab isn’t named, or if searching the name turns up nothing, that’s a serious concern.
- Only HPLC data, no mass spec. As we covered above, HPLC alone confirms purity but not identity. Both tests are standard on a complete COA.
- Identical COAs across different products. If a supplier uses the same COA document for multiple peptides with only the product name swapped, that’s a clear warning sign.
- Suspiciously perfect numbers. Consistent 99.99% purity across every batch? Real lab results vary slightly from batch to batch. Perfect uniformity can signal fabricated data.
Does every missing detail automatically mean fraud? No. But stacking up multiple red flags should make any researcher think twice before trusting the documentation — or the supplier behind it.
Why Does Alpha Peptides Provide Batch-Specific GLP-3 COAs?
Every GLP-3 order from Alpha Peptides includes a batch-specific Certificate of Analysis from an independent third-party laboratory. Each COA contains HPLC purity data, mass spectrometry identity confirmation, the testing lab’s name, and a batch number that matches the label on your vial.
We’ve found that transparency removes guesswork. When a COA is batch-specific and independently produced, you don’t have to take the supplier’s word for it. You can verify the results yourself — and that’s the whole point.
You can browse all current COAs on our Certificates of Analysis page, or go directly to our research-grade GLP-3 product page to see the documentation that ships with every order.
Frequently Asked Questions About GLP-3 COAs
What purity should a GLP-3 COA show?
For research-grade GLP-3, look for HPLC purity of 98% or higher. According to AOAC International validation guidelines, HPLC testing is reproducible above 99% accuracy under controlled conditions (AOAC, 2022). Below 95% purity, impurities may introduce unwanted variables in experimental work. Learn more in our guide on what 99% purity actually means for peptides.
Can I verify the lab listed on my GLP-3 COA?
Yes — and you should. Search the lab’s name online and check for ISO/IEC 17025 accreditation. Accredited labs are independently audited for competence and must meet international standards for testing accuracy. If the lab name doesn’t produce any verifiable results, treat that as a red flag.
What’s the difference between a batch-specific and a generic COA?
A batch-specific COA ties the test results to one particular production lot, identified by a unique batch number. A generic COA has no batch number — it could apply to any lot (or no lot at all). Only batch-specific COAs provide meaningful quality assurance for research, because they confirm the results apply to the exact product you received.
Where can I see Alpha Peptides’ GLP-3 COAs?
All Alpha Peptides COAs are publicly available at alpha-peptides.com/coas/. Each document is issued by an independent third-party lab, includes HPLC and mass spectrometry data, and carries a batch number matching the product vial label. You can also browse our full peptide catalog.
For research use only. Not for human consumption. This article is for informational and educational purposes related to analytical chemistry and laboratory quality assurance. It does not constitute medical advice, dosing guidance, or therapeutic recommendations. GLP-3 is a research compound sold exclusively for use by qualified laboratory professionals.