· For research use only. Not for human consumption.
For research use only. Not for human consumption.
When you see “99% purity” on a vial of GLP-3, what does that number actually tell you? GLP-3 purity is one of the most important quality markers for this triple incretin receptor agonist analog — yet most people never learn what the number means or how it’s measured. A 2021 study in Analytical Chemistry found that even small impurity differences can alter experimental outcomes in receptor-binding assays (Guo D et al., Analytical Chemistry, 2021).
So let’s break it down. No chemistry degree required. If you’ve ever checked an ingredient label at the grocery store, you already have the mental framework you need.
New to this compound? Start with our beginner’s guide to GLP-3 for the basics on what it is and why researchers study it.
TL;DR: GLP-3 purity tells you what percentage of a sample is actually the target compound versus unwanted leftovers from manufacturing. A 99% reading means 1% is something else — typically broken chains or oxidized fragments. Research published in Peptides shows that impurity levels of 3-5% can shift experimental results in receptor-binding studies (Rivier et al., 2018). Always verify purity with a third-party Certificate of Analysis. For research use only. Not for human consumption.
What Does GLP-3 Purity Actually Mean?
According to USP General Chapter <1080>, purity measures the fraction of a sample that consists of the intended compound versus everything else present (United States Pharmacopeia, 2023). For GLP-3, that means how much of what’s in the vial is actually GLP-3 — and how much is leftover manufacturing debris.
Here’s a simple way to think about it. Imagine buying a bottle of orange juice. The label says “100% juice,” but if you read the fine print, it might actually contain 95% real juice and 5% water, pulp, and other fillers. GLP-3 purity works the same way. When the label says 99%, it means 99% of the sample is the actual target peptide. The remaining 1% is something else.
That “something else” matters. In a glass of juice, a little extra water won’t ruin your morning. But in a research experiment, even a tiny amount of the wrong compound can throw off your results entirely.
How Is GLP-3 Purity Measured?

The gold standard for measuring GLP-3 purity is a technique called HPLC — high-performance liquid chromatography. A 2004 review in Methods in Molecular Biology identified HPLC as the primary method for peptide purity testing across pharmaceutical and research applications (Aguilar, Humana Press, 2004). It sounds complicated, but the concept is surprisingly straightforward.
Think of HPLC like a very precise sorting machine. You dissolve a tiny amount of the sample in liquid and push it through a long, narrow tube packed with microscopic particles. Different molecules travel through this tube at different speeds — like runners in a race where some are faster than others.
A detector at the finish line records when each molecule arrives and how much of it there is. The machine plots these results as a series of peaks on a graph called a chromatogram. The tallest peak is your target compound. The small bumps beside it are the impurities.
The math is simple: divide the area of the main peak by the total area of all peaks, multiply by 100, and you get the purity percentage. That’s where “99%” comes from. For a deeper look at how this technology works, check out our guide on what HPLC testing tells you about peptide quality.
What’s Hiding in the Other 1-5%?
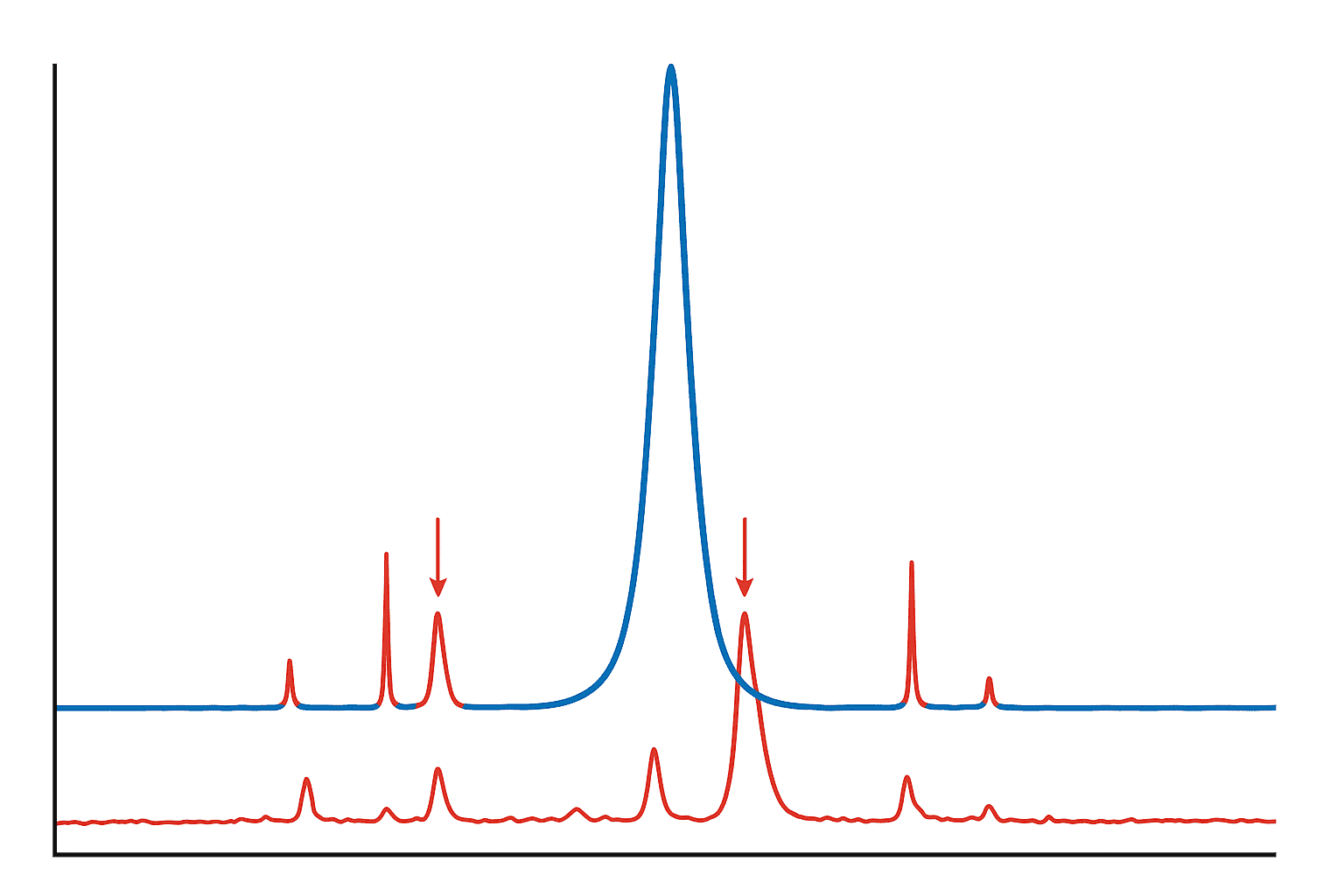
A 2021 analysis in Analytical Chemistry identified four main types of impurities in synthetic peptides: deletion sequences, oxidized species, truncated chains, and leftover manufacturing chemicals (Guo D et al., 2021). Each of these is worth understanding in plain language.
Deletion Sequences — Missing Puzzle Pieces
GLP-3 is a chain of amino acids linked together in a specific order. During manufacturing, these amino acids are added one at a time. Sometimes a step fails and one amino acid gets skipped. The result is a chain that looks almost right but has a gap. It’s like a necklace with a missing bead — close, but not the same piece of jewelry.
Oxidized Fragments — Rust on the Chain
Certain amino acids react with oxygen during production or storage. When that happens, the molecule changes shape slightly. Think of it like rust on a metal tool. The tool still looks like a wrench, but it won’t grip a bolt the same way. An oxidized GLP-3 fragment won’t interact with receptors the way the intact molecule does.
Residual Chemicals — Factory Leftovers
Manufacturing uses solvents and acids to build and purify the peptide. Small traces of these chemicals can stick around in the final product. A 2022 study in the Journal of Peptide Science found that TFA (a common manufacturing acid) alone can account for 10-30% of a peptide sample’s total weight (Zompra AA et al., 2022). These residuals don’t show up on HPLC, which is one reason purity percentage isn’t the full picture.
Why Does the Difference Between 95% and 99% GLP-3 Purity Matter?

Research published in Peptides demonstrated that impurity levels of just 3-5% can measurably alter dose-response curves in receptor-binding assays — exactly the gap between a 95% and 99% sample (Rivier J et al., 2018). That gap isn’t a rounding error. It’s a fivefold difference in the amount of unwanted material.
Let’s put real numbers on it. In a 10 mg vial at 95% purity, you have 500 micrograms of impurities. At 99% purity, you have 100 micrograms. That’s five times less contamination. If any of those impurities happen to interact with the same receptor targets that GLP-3 does, they can quietly skew your experimental data without you realizing it.
Here’s the practical takeaway: for serious receptor-binding or cell-based research, most published studies use compounds at 98% purity or higher. A 95% sample might work for early-stage exploratory work, but it introduces enough noise to undermine precision experiments. Don’t these numbers matter when you’re investing time and resources in research? They really do.
How Can You Verify GLP-3 Purity Claims?

According to USP General Chapter <1080>, a legitimate Certificate of Analysis must include the test method, the result, the specification limit, and the identity of the testing laboratory (USP, 2023). Without all four, the document is incomplete — no matter how impressive the purity number looks.
Here’s what to check on any COA before trusting a GLP-3 purity claim:
- HPLC purity percentage — this is the headline number, but it needs context
- Mass spectrometry confirmation — HPLC tells you how pure; mass spec confirms what’s actually in the vial (see our guide on what mass spectrometry reveals about peptide quality)
- Batch or lot number — ties the results to your specific vial, not a generic template
- Testing laboratory name — should be a third-party lab, not the supplier testing their own product
- Date of analysis — confirms the testing is recent and batch-specific
A COA without a named, independent testing lab is like a restaurant health inspection where the restaurant grades itself. You want an outside party with no financial stake in the result. Purity numbers from in-house testing carry an inherent conflict of interest. For a full walkthrough of COA interpretation, read our guide on what 99% purity actually means for peptides.
Frequently Asked Questions About GLP-3 Purity
What is a good purity level for research-grade GLP-3?
Most published preclinical studies use peptides at 98% purity or higher. A 2018 study in Peptides found that impurity levels below 3% minimize interference in receptor-binding assays (Rivier et al., 2018). For quantitative research, 98%+ is the practical standard. For early exploratory work, 95% can be acceptable, but expect more variability in results.
Can you see GLP-3 purity results before buying?
Reputable suppliers publish batch-specific COAs showing HPLC purity and mass spectrometry results. Alpha Peptides makes these available on the Certificates of Analysis page. If a supplier won’t share testing documentation before purchase, that’s a significant red flag worth paying attention to.
Does GLP-3 purity change over time?
Yes. Peptides can degrade with heat, moisture, and UV light exposure. Oxidation is the most common form of degradation, gradually converting intact molecules into inactive fragments. Proper storage — typically frozen, sealed, and away from light — slows this process considerably. Our peptide storage guide covers best practices.
What’s the difference between purity and potency?
Purity tells you what percentage of a sample is the target compound. Potency tells you how strongly that compound produces a biological effect. A 99% pure sample of a weak peptide is still weak. A 95% pure sample of a potent peptide is still potent — just contaminated. They’re related but measure fundamentally different things.
Where to Find Verified GLP-3 for Research

If you’re sourcing GLP-3 for laboratory research, purity documentation should be the first thing you evaluate. Not marketing claims, not price, not shipping speed — the COA.
Alpha Peptides provides research-grade GLP-3 with batch-specific third-party testing, including HPLC purity analysis and mass spectrometry identity confirmation. Every order ships with cold-chain packaging from our Derry, New Hampshire facility. Browse the full peptide catalog or review testing documentation on our COA page.
For research use only. Not for human consumption. All peptides referenced in this article are research chemicals intended exclusively for laboratory and preclinical research purposes. They are not dietary supplements, are not approved for human or veterinary use, and are not intended to diagnose, treat, cure, or prevent any disease or condition. All information on this page is provided for educational purposes relating to peptide analytical chemistry and laboratory research methods only. It does not constitute medical advice of any kind.




