· For research use only. Not for human consumption.
For research use only. Not for human consumption.
Most peptides are fragile. They break down in heat, degrade in acid, and lose activity when stored improperly. BPC-157 stability, however, is one of the most frequently discussed aspects of this research compound — because it seems to break the rules.
Researchers have noted that BPC-157 resists degradation in conditions that destroy other peptides of similar size. This guide explains why that matters, what makes BPC-157 more durable, and what it means for your research.
For background on this peptide’s origins, see our guide on BPC-157’s gastric juice connection.
TL;DR: BPC-157 stability is unusually high compared to most research peptides. Its resistance to acid degradation, thermal stress, and enzymatic breakdown has been documented in preclinical literature. This resilience likely comes from its high proline content (4 out of 15 amino acids) and its origin in gastric juice — one of the body’s harshest environments. Seiwerth et al. (2021) reviewed stability data across multiple experimental conditions (PMID: 34267654). For research use only. Not for human consumption.
Why Most Peptides Are Fragile
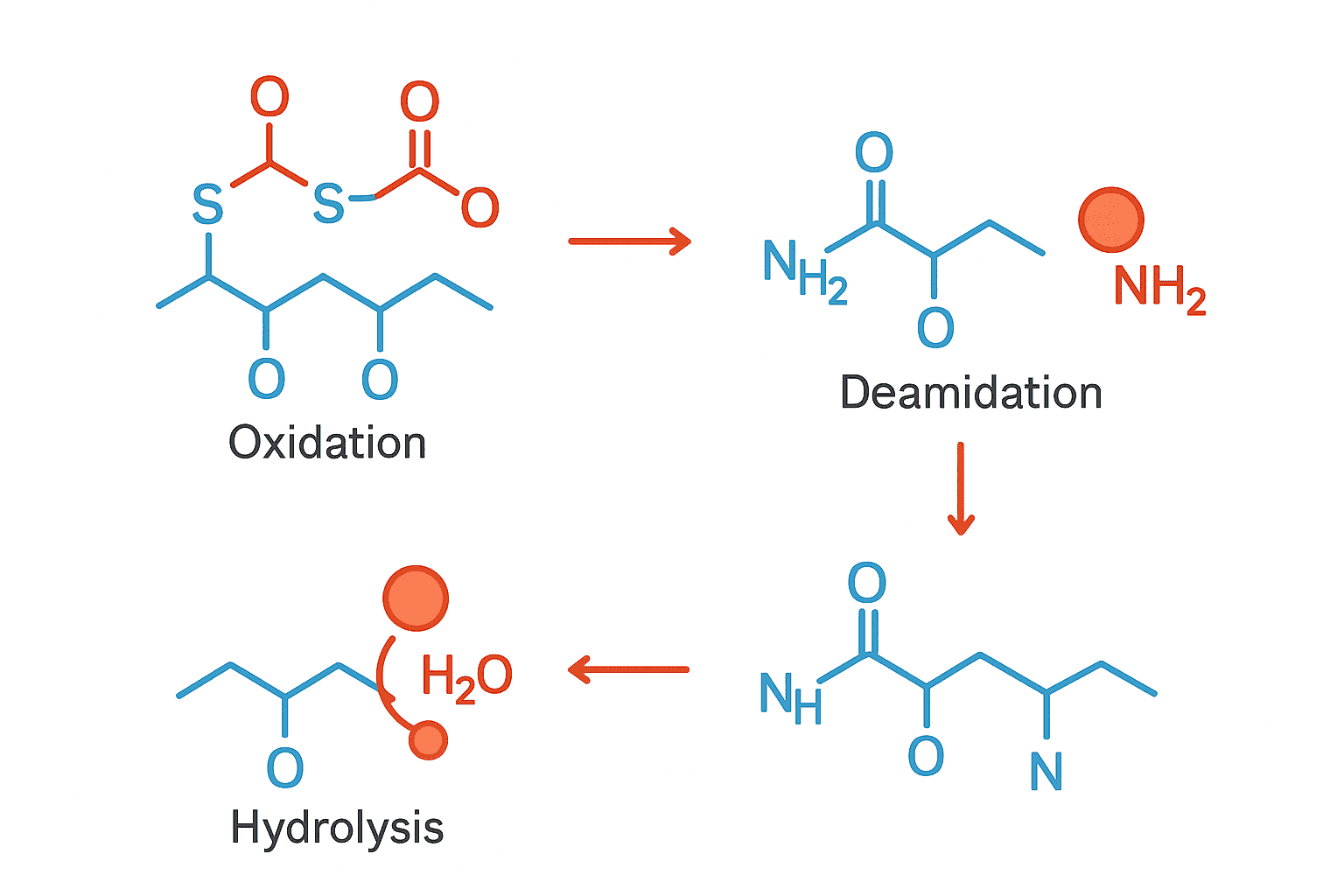
To appreciate BPC-157 stability, you first need to understand why most peptides are delicate. A typical peptide is a chain of amino acids held together by peptide bonds. These bonds are vulnerable to:
- Acid hydrolysis — Acidic conditions can break peptide bonds apart. Your stomach uses this exact mechanism to digest dietary proteins.
- Enzymatic degradation — Enzymes called proteases are specifically designed to cut peptide bonds. They’re everywhere — in blood, in tissue, in digestive fluid.
- Thermal degradation — Heat causes peptides to unfold (denature) and can accelerate chemical reactions that break bonds.
- Oxidation — Exposure to oxygen can modify certain amino acids, changing the peptide’s structure and function.
Most research peptides have a half-life measured in minutes to hours in biological conditions. They need careful handling, cold storage, and protective formulations to remain active.
What Makes BPC-157 Stability Different

Several structural features contribute to BPC-157 stability:
High Proline Content
Four out of BPC-157’s fifteen amino acids are proline — that’s over 26%, which is remarkably high. Proline creates rigid kinks in the peptide chain because its side chain loops back and bonds to the backbone. This rigidity makes it harder for proteases to grab and cut the chain. It’s like trying to cut a wire that keeps bending away from your scissors.
Gastric Origin
BPC-157 was discovered in gastric juice — a fluid with pH as low as 1.5. Any peptide that naturally exists in this environment must resist acid hydrolysis. Evolution (or in this case, the biology of gastric protein production) has selected for a structure that can withstand extreme acidity.
Compact Structure
The combination of proline’s rigidity and the arrangement of charged amino acids (glutamic acid, aspartic acid, lysine) creates a relatively compact three-dimensional structure that doesn’t easily unfold. Since unfolding exposes vulnerable bonds, a compact structure provides inherent protection.
Seiwerth and colleagues (2021) documented BPC-157’s stability across a range of experimental conditions in their review published in Frontiers in Pharmacology, noting its resilience in both acidic and neutral pH environments. (PMID: 34267654)
BPC-157 Stability in Practical Research Settings
For researchers, BPC-157 stability has practical implications:
Storage
Like all research peptides, BPC-157 should be stored lyophilized (freeze-dried) at -20°C for long-term stability. However, its inherent resilience means it may tolerate brief temperature excursions better than more fragile peptides. This doesn’t mean you should be careless — it just means the margin for error is slightly wider.
Reconstitution
Once reconstituted in bacteriostatic water, BPC-157 solutions should still be refrigerated at 2-8°C. The benzyl alcohol in bacteriostatic water provides antimicrobial protection, but the peptide’s own stability helps maintain its integrity in solution.
Experimental Design
BPC-157 stability means researchers can design longer-duration experiments with greater confidence that the peptide remains active throughout. With more fragile peptides, degradation during the experiment itself can confound results.
Seiwerth, Sikiric, and colleagues (1997) published early characterization data on BPC-157’s resistance to gastric acid degradation, establishing the foundation for subsequent stability research. (PMID: 9403790)
Comparing BPC-157 Stability to Other Peptides
To put BPC-157 stability in context, consider how it compares to other research peptides:
- Most linear peptides (5-20 amino acids) — Half-life in serum measured in minutes. Rapidly degraded by proteases.
- Cyclic peptides — Ring structures provide enhanced stability. BPC-157 isn’t cyclic but achieves similar resilience through proline content.
- Modified peptides (PEGylated, D-amino acid substitutions) — Chemical modifications can dramatically increase stability, but BPC-157 achieves notable durability without any modifications to its natural sequence.
This natural resilience, without requiring chemical modifications, is part of what makes BPC-157 interesting to researchers. Alpha Peptides’ BPC-157 + TB-500 is tested for purity and identity — see our COA page for batch-specific results.
Frequently Asked Questions
How stable is BPC-157 compared to other peptides?
BPC-157 stability is significantly higher than most linear peptides of similar size. Its resistance to acid degradation and enzymatic breakdown has been documented in multiple preclinical studies.
Does stability mean BPC-157 doesn’t need cold storage?
No. While BPC-157 is more resilient than many peptides, proper cold storage (-20°C lyophilized, 2-8°C reconstituted) is still recommended to maximize shelf life and maintain research-grade quality.
Why does proline make peptides more stable?
Proline’s ring structure creates rigid kinks that resist enzymatic cleavage. Proteases (enzymes that cut peptide bonds) have difficulty accessing bonds adjacent to proline residues, making proline-rich peptides harder to degrade.
Can BPC-157 survive in stomach acid?
Preclinical research suggests BPC-157 maintains its structure in highly acidic conditions similar to gastric fluid. This is consistent with its origin — it was discovered in gastric juice, an environment with pH as low as 1.5.
For research use only. Not for human consumption. This article is intended for informational purposes and does not constitute medical advice, dosing guidance, or therapeutic recommendations.