· For research use only. Not for human consumption.
For research use only. Not for human consumption.
You see it everywhere: “99% purity,” “98% purity,” “95%+ purity.” Every peptide supplier puts a number on the label. But what does purity actually mean for a research peptide, and does the difference between those numbers actually matter?
It matters more than most people realize — and in a different way than you might expect. Purity isn’t a marketing grade. It’s an analytical measurement with real implications for research validity. This post breaks down exactly what that number represents, what’s hiding in the remaining 1–5%, and what to look for when you’re reading a certificate of analysis.
TL;DR: Peptide purity is measured by HPLC and represents what percentage of the sample is actually your target compound. A 99% purity reading means roughly 1% of the vial’s contents are something else — typically deletion sequences, oxidized species, or counter-ions from synthesis. For research purposes, that distinction matters more than most supplier pages explain. For research use only. Not for human consumption.
How Is Peptide Purity Measured?
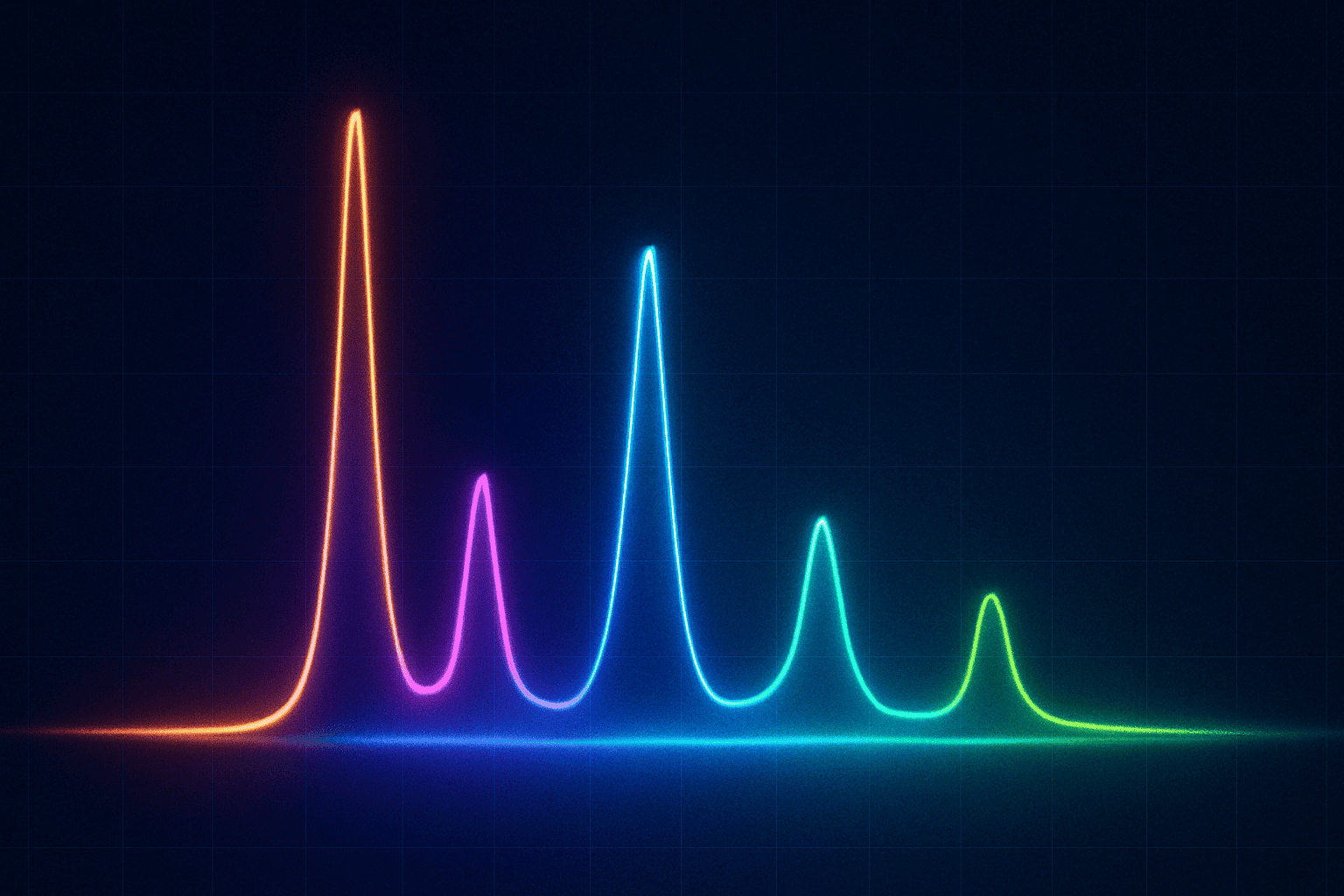
Purity in research peptides is measured by high-performance liquid chromatography, or HPLC — the analytical gold standard for this class of compound. According to a 2019 review in the Journal of Pharmaceutical and Biomedical Analysis, HPLC is the primary technique used by pharmaceutical and research-grade peptide manufacturers to determine compound purity, and it accounts for more than 80% of all purity testing in peptide synthesis workflows. ([Vlasák J et al., Journal of Pharmaceutical and Biomedical Analysis](https://pubmed.ncbi.nlm.nih.gov/31078011/), 2019)
Here’s the simple version of how it works. The HPLC machine pushes a dissolved sample through a column packed with tiny particles. Different molecules travel through at different speeds depending on their size and chemistry. When they exit the column, a detector measures the signal and plots it as a series of peaks on a chromatogram — one peak per distinct compound.
The purity percentage comes directly from those peaks. The machine calculates the “area under the curve” for each one. Your target peptide produces the largest peak. Divide that peak’s area by the total area of all peaks, multiply by 100, and that’s your purity number. A result of 99% means 99% of what’s in the vial, by area, is the target compound.
That last point is important. Purity by HPLC area is not the same as purity by mass. It’s a relative measurement. Two different impurity profiles can both produce a 99% reading while differing significantly in their composition. The chromatogram itself — not just the headline number — is what researchers should ask to see.
HPLC-based purity analysis measures the area-under-the-curve contribution of each compound present in a peptide sample. The target peptide’s peak area, divided by total peak area across the chromatogram, produces the reported purity percentage. A 2019 review in the Journal of Pharmaceutical and Biomedical Analysis identified HPLC as the primary purity-testing method in more than 80% of peptide synthesis quality workflows. (PMID: 31078011)
What Are the Other 1–5%?
If 99% of the vial is your peptide, something accounts for the remaining 1%. Understanding what that something is matters for research design. A 2021 paper in Analytical Chemistry identified four primary categories of impurities in synthetic peptides: deletion sequences, oxidized species, truncated or scrambled sequences, and residual synthesis reagents — each with distinct potential effects on biological assays. ([Guo D et al., Analytical Chemistry](https://pubmed.ncbi.nlm.nih.gov/33439629/), 2021)
Deletion Sequences
These are the most common impurity category. During solid-phase peptide synthesis, amino acids are added one at a time. If a coupling step doesn’t go to completion, the next amino acid gets added anyway — producing a slightly shorter peptide with one residue missing. That truncated chain is called a deletion sequence. It may be chemically similar to your target compound but it’s structurally distinct, and in receptor-binding assays, that distinction can produce unexpected signals.
Oxidized Species
Some amino acids — methionine and cysteine particularly — are prone to oxidation during synthesis or storage. An oxidized version of your peptide is a different molecule. It won’t behave the same way in an experiment. The extent of oxidation depends on synthesis conditions, storage environment, and how long the compound has been sitting before use.
TFA Counter-Ions
Most peptide synthesis uses trifluoroacetic acid (TFA) as a cleavage agent and solvent during purification. TFA doesn’t always wash out completely. Residual TFA counter-ions attach to the peptide salt and become part of the gross weight without contributing to net peptide content. This matters beyond the purity percentage — we’ll come back to it in a moment.
Moisture and Residual Solvents
Lyophilized peptides absorb moisture from the environment. Water content contributes to gross weight without being peptide. Residual acetonitrile and other solvents from the purification process can also persist in small amounts. None of these show up as distinct peaks on an HPLC chromatogram — they fall outside what HPLC measures, which is one reason purity percentage and actual peptide content aren’t the same thing.
Does the Difference Between 95% and 99% Matter for Research?
Yes — and it matters most in exactly the situations where you’d least want ambiguity. Research published in Peptides by Rivier and colleagues demonstrated that impurity profiles as low as 3–5% can measurably shift dose-response curves in receptor-binding assays, particularly when the impurity shares partial structural homology with the target peptide. ([Rivier J et al., Peptides](https://pubmed.ncbi.nlm.nih.gov/29763693/), 2018)
Think about what that means in practice. You’re running an in vitro experiment with a 95%-pure compound. Five percent of what you add to the assay is something other than your target. If even a fraction of that 5% binds to the same receptor — perhaps with different affinity — your dose-response curve shifts. Your EC50 looks different than it should. You reproduce the experiment with a new batch, and if the impurity profile has changed, your results don’t replicate cleanly.
Reproducibility is the core issue. Science depends on being able to repeat a result. When compound purity varies between batches — or between suppliers — it introduces a variable that’s hard to control for and easy to overlook. That’s why many published preclinical studies specify the purity of the compounds used. It’s not a formality. It’s a methodological control.
The practical threshold most researchers use is 98%+ for receptor-binding or cell-based work. 95% is acceptable for some early exploratory studies where precision matters less, but it should never be treated as equivalent to 99%. The two numbers represent meaningfully different sample compositions.
The gap between 95% and 99% purity isn’t just a 4-point quality score — it represents a four- to fivefold increase in the absolute quantity of impurities present in every experiment. In a 10 mg vial, that’s 100-200 additional micrograms of unknown compounds potentially active in your assay.
A 2018 study in Peptides by Rivier and colleagues found that impurity profiles of 3–5% in synthetic peptides can measurably alter dose-response curves in receptor-binding assays, particularly when impurities share partial structural homology with the target compound. The authors recommended using compounds of 98% purity or greater in assays designed to generate quantitative binding data. (PMID: 29763693)
What About Gross Weight vs. Net Peptide Content?
Here’s the distinction that almost never gets explained on supplier pages, and it’s the one that most directly affects your research budget. Gross weight is what’s in the vial. Net peptide content is how much of that is actually your peptide. A 2022 analysis in the Journal of Peptide Science found that TFA counter-ion content alone can account for 10–30% of a lyophilized peptide’s gross weight, depending on the compound’s amino acid composition and the supplier’s wash protocols. ([Zompra AA et al., Journal of Peptide Science](https://pubmed.ncbi.nlm.nih.gov/35322530/), 2022)
So the label says 10 mg. What’s actually in the vial?
If net peptide content is 75%, you’re working with 7.5 mg of actual compound. The rest is TFA salts, moisture, and residual solvents. This matters enormously when you’re calculating concentrations for an experiment. Use gross weight to calculate your solution concentration and every downstream measurement is off.
Some suppliers report net peptide content on their COA. Many don’t. The ones who don’t are either unaware of why it matters or choosing not to highlight a number that makes their product look less valuable. Either way, it’s information you should have. Calculating net peptide content requires knowing the peptide’s molecular weight, its TFA content (measurable via ion chromatography or NMR), and its moisture content (measured by Karl Fischer titration).
If a COA doesn’t report net peptide content separately from HPLC purity, that’s a gap worth asking about. The two numbers are related but they’re not the same, and conflating them is one of the most common sources of experimental error in peptide research.
Researchers who switch from sourcing by gross weight to sourcing by verified net peptide content frequently find their experimental concentrations shift by 15–25% — not because the compound changed, but because they finally know what’s actually in the vial.
What to Look for on a COA: Reading Purity Claims Correctly
A COA (certificate of analysis) is the document that should back up any purity claim a supplier makes. According to the United States Pharmacopeia (USP), a legitimate analytical COA for a research compound must include the test method used, the result obtained, the specification limit, and the identity of the testing laboratory. ([United States Pharmacopeia, General Chapter <1080>](https://www.usp.org/), 2023) Without all four elements, the document is incomplete.
What a Legitimate COA Should Show
At minimum, look for an HPLC purity percentage with the method specified (typically reversed-phase HPLC with UV detection at 220 nm). Look for a mass spectrometry result confirming the compound’s molecular weight matches the known structure of the target peptide. Look for the testing lab’s name — and verify it’s an independent third party, not the supplier’s own facility.
What the HPLC Percentage Does and Doesn’t Tell You
The HPLC percentage tells you what fraction of the sample’s UV-absorbing content is your target compound. It doesn’t tell you the absolute quantity of peptide in the vial. It doesn’t account for moisture, TFA salts, or non-UV-absorbing impurities. For most research applications, HPLC purity combined with mass spec identity confirmation is sufficient — but for quantitative assays, net peptide content data adds another layer of reliability.
Red Flags on a COA
Be cautious of COAs without a test date, COAs that show only a purity percentage with no underlying chromatogram data available, and any document where the testing laboratory is the same entity as the supplier. Purity claims backed by in-house testing lack the independence that makes the number scientifically credible. Third-party testing is the standard.
Alpha Peptides COAs are produced by independent third-party analytical laboratories and include HPLC purity data, mass spectrometry confirmation, and the specific test date for each batch. Full COA documentation is available at alpha-peptides.com/coas/.
Frequently Asked Questions About Peptide Purity
Is 95% pure peptide good enough for research?
It depends on the application. For early exploratory or qualitative studies, 95% purity is often workable. For receptor-binding assays, dose-response work, or any experiment requiring quantitative precision, 95% introduces enough impurity mass to meaningfully affect results. A 2018 study in Peptides found that 3–5% impurity levels can shift dose-response curves in receptor assays — which is exactly what sits between a 95% and 99% reading. ([Rivier J et al., Peptides](https://pubmed.ncbi.nlm.nih.gov/29763693/), 2018) Most published preclinical studies use 98%+ as the standard.
What is a deletion sequence?
A deletion sequence is a truncated version of the target peptide — the same compound with one or more amino acids missing. They form during solid-phase peptide synthesis when a coupling step doesn’t go to completion and the chain continues building from an incomplete intermediate. Deletion sequences are structurally similar to the target compound but chemically distinct. In receptor-binding experiments, they can produce off-target signals that complicate data interpretation.
What is net peptide content?
Net peptide content is the percentage of a peptide sample that is actually the target compound, accounting for TFA counter-ions, moisture, and residual solvents that contribute to gross weight. HPLC purity doesn’t capture these — it only measures relative UV-absorbing content. A 2022 paper in the Journal of Peptide Science found TFA salts alone can account for 10–30% of gross weight in lyophilized peptides. ([Zompra AA et al., Journal of Peptide Science](https://pubmed.ncbi.nlm.nih.gov/35322530/), 2022) Net peptide content gives you the real quantity for experimental calculations.
Why do purity levels vary between batches?
Peptide synthesis is a multi-step chemical process, and small variations in reagent quality, reaction conditions, coupling efficiency, and purification yield produce batch-to-batch variation. Even with the same protocol, a synthesis run that encounters slightly different temperature or moisture conditions can produce a subtly different impurity profile. That’s why reputable suppliers test every batch independently — and why the COA should carry a specific lot number tied to its data, not a generic template figure.
The Number on the Label Is a Starting Point, Not the Full Story
99% purity means something real — it means HPLC analysis found that 99% of the UV-absorbing content in the sample is your target compound. That’s meaningful. It’s also not the whole picture.
The remaining 1% has a composition that matters. Gross weight isn’t the same as net peptide content. And a purity percentage without a COA from an independent lab is just a number on a webpage. Researchers who understand these distinctions run tighter experiments and get more interpretable results.
If you’re evaluating a peptide supplier, start with the COA. Ask for the chromatogram, not just the percentage. Verify the testing lab is independent. Check whether net peptide content is reported. Those four steps tell you more about a supplier’s quality standards than any marketing claim will.
Related reading: What Is a Certificate of Analysis (COA)? A Researcher’s Guide | Why Cheap Peptides Are a Research Risk
For research use only. Not for human consumption. All information on this page is provided for educational and scientific reference purposes relating to laboratory and preclinical peptide research. It does not constitute medical advice and should not be interpreted as guidance for personal use. Peptide purity specifications discussed here apply to research settings only.
Shop Research-Grade Peptides from Alpha Peptides
Alpha Peptides is a U.S.-based research peptide supplier providing HPLC-verified, third-party tested compounds with full Certificates of Analysis on every batch. All products are for research use only, not for human consumption.
- BPC-157 — 15-amino acid peptide, >98% HPLC purity
- TB-500 — Thymosin Beta-4 fragment, MS-confirmed identity
- Ipamorelin — Selective GHS-R agonist, research grade
- CJC-1295 (with DAC) — GHRH analog with full analytical documentation
Browse all 20+ research peptides → | View Certificates of Analysis →