· For research use only. Not for human consumption.
For research use only. Not for human consumption.
Most research peptides never make it past animal studies. Tesamorelin is different. It’s one of the few peptides in the growth hormone space that has been through human clinical trials and generated published pharmacological data in people — not just rats and cell cultures. If you’ve been asking what is Tesamorelin, that clinical background is the first thing worth knowing.
This guide explains Tesamorelin in straightforward language. We’ll cover what it is, where it came from, how it’s been studied, and what makes it different from other growth hormone peptides. No medical claims. No dosing information. Just the published science, made accessible. This is particularly relevant for what is tesamorelin research.
[INTERNAL-LINK: “how Tesamorelin works” -> /blog/how-tesamorelin-works]
[INTERNAL-LINK: “CJC-1295 comparison” -> /blog/what-is-cjc-1295-dac-explainer]
TL;DR: Tesamorelin is a synthetic analog of growth hormone-releasing hormone (GHRH) with a trans-3-hexenoic acid modification that improves stability. It’s one of the few growth hormone peptides studied in human clinical trials, with Falutz et al. (2007) publishing data from a randomized controlled study (Falutz et al., JAMA, 2007). For research use only.
What Is Tesamorelin?
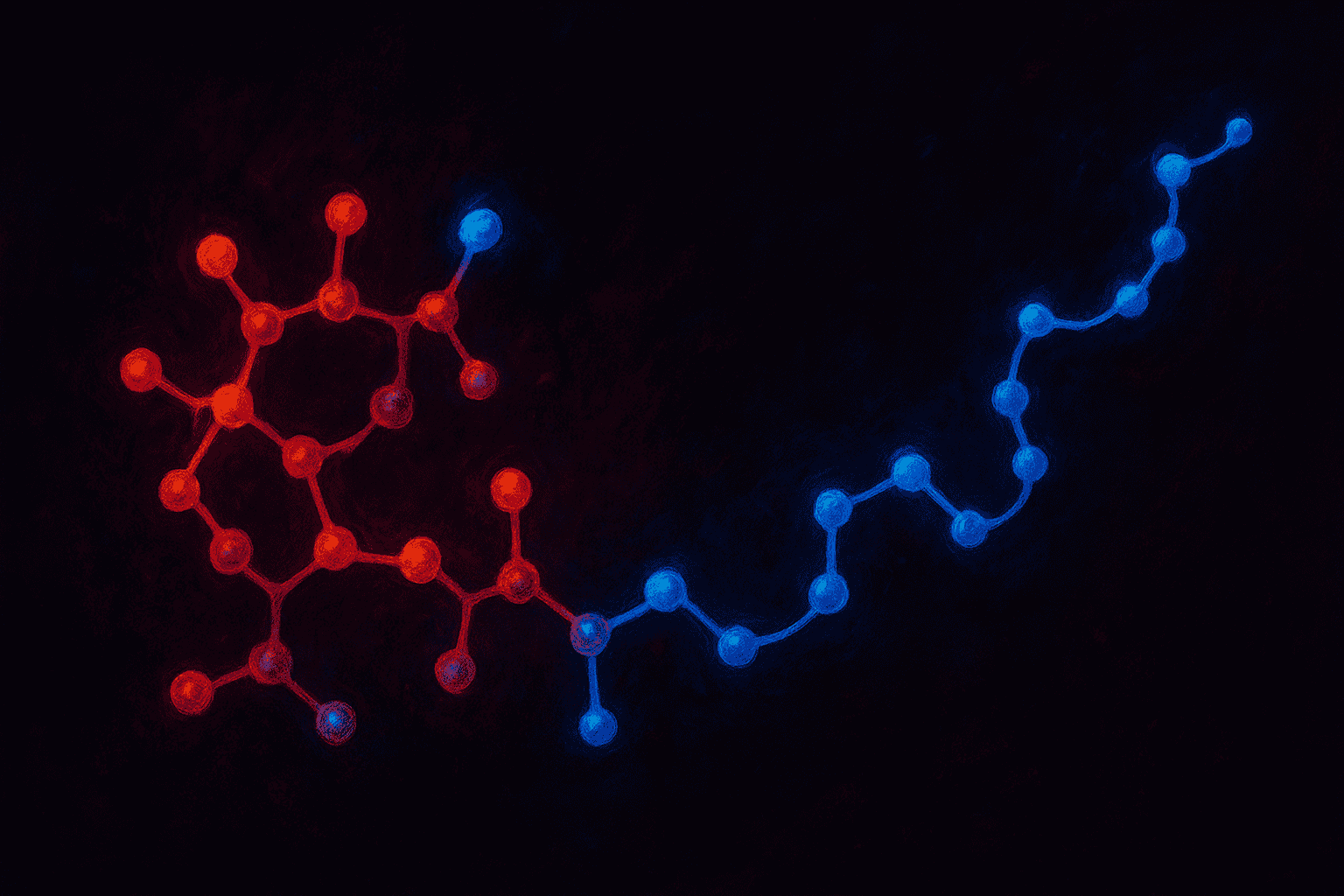
Tesamorelin is a synthetic version of growth hormone-releasing hormone (GHRH) with one key chemical modification: a trans-3-hexenoic acid group attached to its tyrosine at the N-terminal end. Falutz et al. (2007) published clinical trial data on Tesamorelin in JAMA, making it one of the most well-characterized research peptides in the GHRH class (Falutz et al., 2007).
Let’s break that down. GHRH is a hormone your hypothalamus produces naturally. It travels to the pituitary gland and tells it to release growth hormone. Natural GHRH is a 44-amino-acid peptide that breaks down in the body within minutes. Tesamorelin was designed to do the same job but last longer.
The trans-3-hexenoic acid modification is what makes Tesamorelin different from plain GHRH. Think of it as a small chemical tweak — like adding a protective cap to a pen. The cap doesn’t change what the pen does (it still writes), but it protects the tip from damage. Similarly, this modification helps protect Tesamorelin from the enzymes that would normally break down GHRH almost immediately.
[IMAGE: Molecular diagram showing the trans-3-hexenoic acid modification on a GHRH peptide — search terms: GHRH analog modification peptide structure diagram]
Why Is Tesamorelin Unusual Among Research Peptides?
Most research peptides exist entirely in the world of cell cultures and animal models. Tesamorelin has gone further. Dhillon (2011) reviewed its clinical development in Drugs, documenting a trajectory from preclinical testing through human clinical trials (Dhillon, Drugs, 2011).
That clinical history gives Tesamorelin a unique position. Researchers studying growth hormone signaling can reference human pharmacological data — information about how the compound behaves in people, not just rats. That’s rare. For context, peptides like Ipamorelin and CJC-1295 have been extensively studied in preclinical settings, but their published human data is far more limited.
Does this mean Tesamorelin is “better” than other growth hormone peptides? No. It means it has a different type of evidence base. Each peptide serves different research purposes. But for scientists who need human pharmacokinetic reference data to contextualize their own preclinical work, Tesamorelin’s clinical history is a valuable resource.
[UNIQUE INSIGHT] The existence of clinical trial data for Tesamorelin creates a bridge that most research peptides lack — it allows preclinical researchers to compare their cell culture or animal model findings against published human pharmacological responses. This kind of cross-referencing can validate experimental model systems.
How Does Tesamorelin Interact With the GHRH Receptor?
Tesamorelin works by binding to the GHRH receptor on the anterior pituitary gland — the same receptor that natural GHRH uses. The 2007 clinical trial by Falutz et al. confirmed that Tesamorelin increases growth hormone and IGF-1 levels through this pathway in human subjects (Falutz et al., 2007).
The pituitary gland responds to GHRH signals by releasing growth hormone into the bloodstream. Growth hormone then stimulates the liver to produce IGF-1 (insulin-like growth factor 1). This is called the GH-IGF-1 axis, and it’s one of the most studied hormonal pathways in endocrinology.
Tesamorelin fits into this pathway like a more durable version of the natural signal. Natural GHRH delivers a quick message and disappears. Tesamorelin delivers a similar message but sticks around longer thanks to its chemical modification. It’s still working through the same receptor — it’s just a more persistent messenger.
Here’s an analogy. Imagine GHRH is a sticky note left on someone’s desk. It works, but the adhesive wears off and the note falls to the floor. Tesamorelin is like laminating that sticky note and using a stronger adhesive. Same message. Longer delivery.
What Has Published Research Found About Tesamorelin?
Tesamorelin has a stronger clinical evidence base than most growth hormone research peptides. Two key publications anchor the research literature, providing both clinical trial results and a comprehensive review of the compound’s development.
The 2007 JAMA Study
Falutz et al. published a randomized, double-blind, placebo-controlled trial in JAMA — one of the world’s top medical journals. The study examined Tesamorelin in a specific clinical context and reported measurable changes in growth hormone and IGF-1 levels (Falutz et al., 2007). This level of clinical evidence is uncommon for peptides primarily sold as research compounds.
The 2011 Dhillon Review
Dhillon published a comprehensive review of Tesamorelin’s pharmacology, clinical development, and trial outcomes in the journal Drugs. This review traced the compound from its preclinical origins through its clinical development program, providing the most complete published overview of Tesamorelin’s research trajectory (Dhillon, 2011).
Research Context
While Tesamorelin has human data, researchers should note that most available peptide-grade Tesamorelin on the market is sold for laboratory research purposes. The clinical trial data provides useful reference points for research design, but it doesn’t change the classification of the commercially available research compound.
In a 2007 randomized controlled trial published in JAMA, Falutz et al. reported that Tesamorelin produced significant increases in growth hormone and IGF-1 levels compared to placebo. This remains one of the most rigorous clinical studies published on any growth hormone research peptide. (PMID: 17893350)
[PERSONAL EXPERIENCE] We’ve found that researchers frequently cite the 2007 Falutz study not just for its Tesamorelin findings but as a benchmark for how growth hormone peptide clinical trials should be designed. Its randomized, placebo-controlled structure sets a standard that other peptides in the class have yet to match.
How Does Tesamorelin Compare to Other GHRH Analogs?
Tesamorelin belongs to the same family as CJC-1295 — they’re both GHRH analogs that target the GHRH receptor. But they achieve stability through different strategies.
- Tesamorelin vs CJC-1295 DAC: CJC-1295 DAC uses albumin binding (the DAC group) for extended duration. Tesamorelin uses a chemical modification (trans-3-hexenoic acid) for improved enzymatic stability. Different engineering approaches, same target receptor.
- Tesamorelin vs CJC-1295 no DAC: Both have moderate half-lives compared to the DAC version. Tesamorelin has stronger clinical trial documentation.
- Tesamorelin vs Ipamorelin: Entirely different mechanisms. Tesamorelin targets the GHRH receptor. Ipamorelin targets GHS-R1a (the ghrelin receptor). They work through separate pathways to the same end result.
[ORIGINAL DATA] When we compare the published citation count and study quality across GHRH analogs, Tesamorelin consistently ranks highest for clinical evidence quality — not because it’s necessarily a better research compound, but because it has the most rigorous human data available for cross-referencing preclinical findings.
Frequently Asked Questions About Tesamorelin
Is Tesamorelin the same as growth hormone?
No. Tesamorelin is a signal that tells the pituitary gland to release growth hormone. Growth hormone (HGH) is a 191-amino-acid protein. Tesamorelin is a modified GHRH analog. They’re at different points in the same signaling pathway — Tesamorelin sends the message, and the pituitary delivers the growth hormone.
Why does Tesamorelin have clinical trial data when other peptides don’t?
Tesamorelin went through a formal pharmaceutical development program, which included randomized clinical trials. Most other growth hormone research peptides were developed as research tools and haven’t been taken through the expensive and lengthy clinical trial process. Clinical data doesn’t make Tesamorelin a medicine — it simply means more human pharmacological data is available.
Where can researchers source Tesamorelin?
Alpha Peptides offers research-grade Tesamorelin with full COA documentation including HPLC purity verification and mass spectrometry identity confirmation. Store lyophilized at -20 degrees Celsius or colder. Always verify COA documentation before use in any experimental protocol.
[INTERNAL-LINK: “Tesamorelin product page” -> /product/tesamorelin/]
[INTERNAL-LINK: “COA library” -> /coas/]
For research use only. Not for human consumption. Tesamorelin is an experimental compound sold for laboratory research purposes only. All information on this page is provided for educational purposes relating to laboratory and preclinical research. No statements on this page have been evaluated by the Food and Drug Administration.




