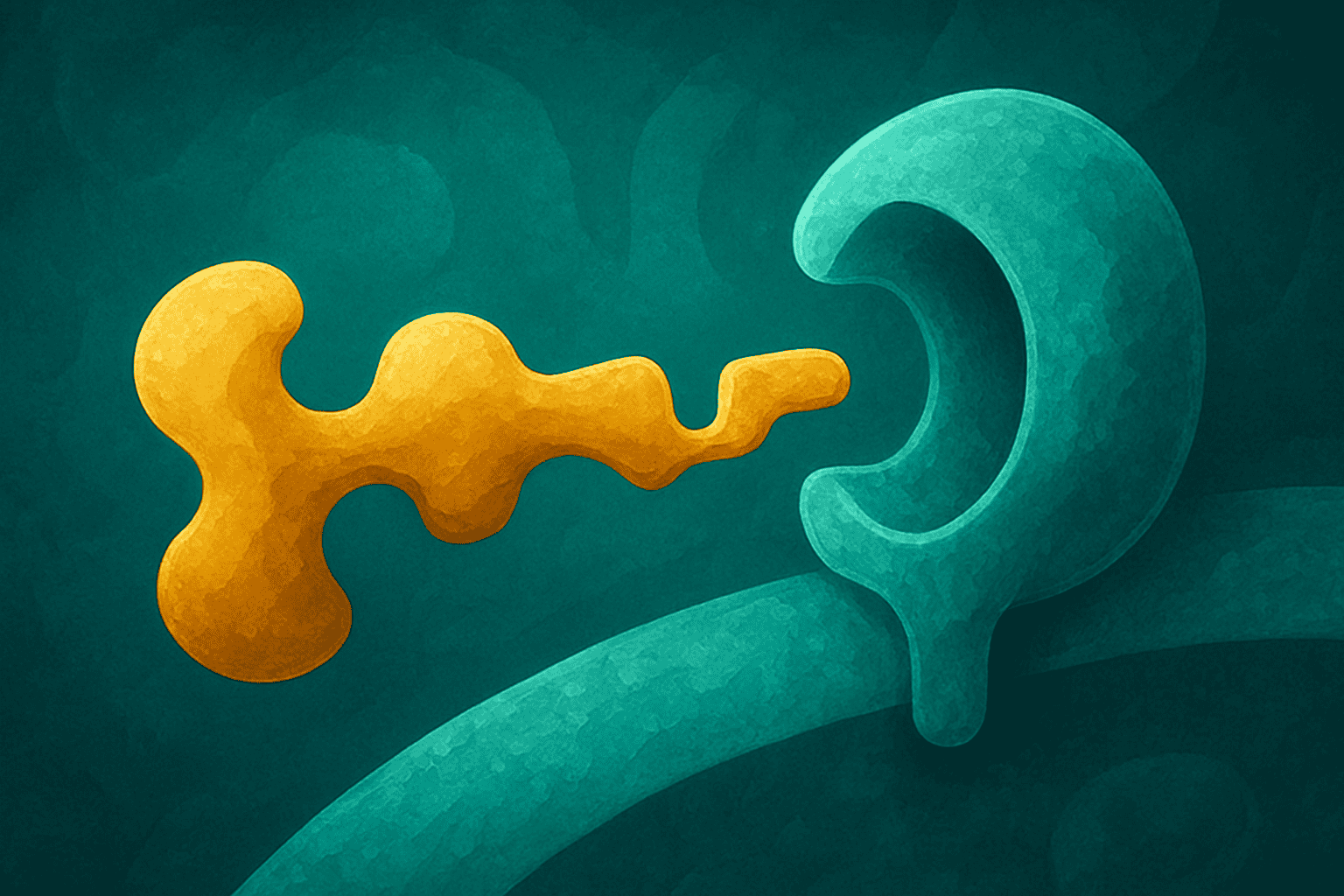· For research use only. Not for human consumption.
For research use only. Not for human consumption.
Peptide receptor binding is the foundation of how research peptides work at the cellular level. When a peptide arrives at a cell, it does not just crash into the membrane and hope for the best. It finds a specific receptor — a protein sitting on the cell surface — and fits into it like a key sliding into a lock. That interaction is what triggers everything that happens next inside the cell.
This lock-and-key concept sounds simple, and at its core, it is. But the details of how tightly a peptide fits, how the receptor changes shape during binding, and what signals get sent afterward are what make peptide receptor binding such a rich area of research. Every research peptide in the catalog interacts with cells through some version of this process.
This guide explains peptide receptor binding in plain language. No biochemistry degree required. For examples of how specific peptides interact with receptors, see our receptor agonist guide or our overview of triple agonist research.
TL;DR: Peptide receptor binding is how peptides communicate with cells. The peptide (key) fits into a receptor protein (lock) on the cell surface, triggering a signal inside the cell. Binding affinity measures how tightly the key fits. Specificity determines which locks a key can open. The induced fit model shows that both the key and lock change shape slightly during binding. For research use only. Not for human consumption.
The Lock-and-Key Model of Peptide Receptor Binding
The lock-and-key model is the simplest way to understand how peptide receptor binding works. Proposed by Emil Fischer over a century ago, this model says that a molecule (the key) has a shape that precisely matches the shape of a receptor (the lock). Only the right key opens the right lock.
In biological terms, receptors are proteins embedded in the cell membrane. They have a binding site — a pocket or groove with a specific three-dimensional shape. When a peptide with the matching shape arrives, it slides into that pocket. The physical and chemical properties of the peptide’s amino acid side chains interact with the amino acids lining the receptor pocket. Hydrogen bonds form. Electrostatic charges attract. The peptide locks into place.
Once locked in, the receptor changes its behavior. It might open a channel in the membrane, activate an enzyme inside the cell, or trigger a chain of chemical reactions called a signaling cascade. The cell receives the peptide’s “message” through this process. Without binding, no message gets delivered.
This is why amino acid sequence matters so much in peptide research. Gwyer et al. (2019) reviewed preclinical studies on BPC-157, a 15-amino-acid peptide whose specific sequence determines which receptors it can interact with (PMID: 30915550). Change the sequence, and you change which locks the key can open.
The Induced Fit Model: When Both Lock and Key Change Shape
The original lock-and-key model is useful but not quite complete. In reality, both the peptide and the receptor change shape slightly when they interact. This more accurate model is called the induced fit model, proposed by Daniel Koshland in the 1950s.
Think of a handshake. When two people shake hands, both hands adjust. Fingers wrap around. Grips tighten. Neither hand stays perfectly rigid — they mold to each other. That is induced fit. The receptor adjusts its shape to better accommodate the peptide, and the peptide may adjust its conformation to better sit in the binding pocket.

This concept matters for peptide receptor binding research because it explains why some peptides that do not look like a perfect match on paper can still bind effectively. The receptor is not a rigid lock — it has some flexibility. A peptide that is close enough in shape can still trigger binding because the receptor bends to accommodate it.
The induced fit model also explains why peptide modifications can fine-tune binding. When researchers add a D-amino acid substitution or attach a chemical group to a peptide, they are adjusting the key’s shape. Sometimes a small change makes the fit tighter (higher affinity). Sometimes it makes the fit looser or blocks binding entirely. Understanding induced fit is what allows scientists to engineer better research compounds.
Binding Affinity: How Tightly Does the Key Fit?
Not all keys fit their locks equally well. Some peptides bind to their receptors very tightly and stay locked in for a long time. Others bind loosely and detach quickly. This strength of interaction is called binding affinity.
Binding affinity is measured in the laboratory using specialized assays. Scientists express it as a dissociation constant (Kd) — the lower the Kd value, the tighter the binding. A peptide with a Kd of 1 nanomolar binds very tightly. One with a Kd of 1 micromolar binds a thousand times less tightly.
Why does affinity matter? Because it determines how much of a peptide is needed to produce a measurable effect in an experimental system. A high-affinity peptide produces a response at very low concentrations. A low-affinity peptide requires much more material to achieve the same level of receptor occupancy. For researchers designing experiments, this directly affects how much compound they need and how they interpret their results.
SS-31, for example, was specifically engineered for high affinity to cardiolipin on the inner mitochondrial membrane. Mitchell et al. (2020) documented this targeted interaction in aged mouse models (PMID: 32273339). The peptide’s four amino acids were deliberately chosen to maximize binding strength at that specific target.
Mitchell et al. (2020) investigated SS-31’s interaction with the inner mitochondrial membrane in aged mouse skeletal muscle, demonstrating that this synthetic tetrapeptide’s engineered amino acid sequence produces specific binding interactions with cardiolipin-associated protein complexes. Published in eLife. (PMID: 32273339)
Specificity: Why a Peptide Activates Some Receptors but Not Others
Specificity is the concept that explains why a given peptide interacts with certain receptors while ignoring others entirely. It is the selectivity of the key. A house key opens your front door but does not open your car, your office, or your neighbor’s house. Peptide specificity works the same way.
Specificity arises from the precise arrangement of amino acid side chains on the peptide. Each side chain has particular chemical properties — some are positively charged, some are negatively charged, some are oily, some attract water. The receptor’s binding pocket has a complementary arrangement. Only a peptide whose side chain pattern matches the pocket’s pattern will fit well enough to bind and activate the receptor.
High specificity is generally desirable in research because it produces cleaner data. If a peptide activates only one receptor type, researchers can confidently attribute any observed effects to that single receptor. If a peptide activates multiple receptors (low specificity), it becomes harder to determine which receptor is responsible for which effect. This is why researchers value highly selective research compounds — they simplify experimental interpretation.
Some peptides are deliberately designed with lower specificity. GLP-3, for instance, is studied as a triple agonist precisely because it activates three receptors. In that case, the multi-receptor interaction is the point of the research, not a limitation.
Signal Transduction: What Happens After the Key Turns
Peptide receptor binding is not the end of the story — it is the beginning. Once a peptide binds to its receptor, a chain of events unfolds inside the cell. This process is called signal transduction. The binding event at the cell surface gets translated into a series of chemical reactions inside the cell that ultimately change the cell’s behavior.
Think of it like a row of dominoes. The peptide binding is the finger that pushes the first domino. That first domino knocks over the second, which knocks over the third, and so on until the last domino falls — which might be a gene getting turned on, a protein getting produced, or a channel opening up. The peptide never enters the cell. It just pushes the first domino.

Different receptors connect to different signaling cascades. That is why the same binding mechanism — key fits lock — can produce vastly different outcomes depending on which receptor is involved. Lee et al. (2015) showed that MOTS-c interacts with the AMPK signaling pathway (PMID: 25738459). Dolotov et al. (2006) documented Semax’s effects on BDNF expression pathways (PMID: 16996037). Same general process of binding and signaling, completely different downstream results.
Understanding peptide receptor binding is essential for interpreting preclinical research data. Alpha Peptides provides research-grade peptides with verified identity and purity so researchers can study these interactions with confidence. Browse the full research catalog or review testing documentation on our Certificates of Analysis page.
Frequently Asked Questions
Does a peptide enter the cell when it binds to a receptor?
In most cases, no. The peptide binds to a receptor on the cell surface and triggers a signaling cascade inside the cell without ever crossing the membrane. The receptor acts as an intermediary, translating the external binding event into an internal chemical response. Some small peptides like SS-31 are exceptions — they can cross membranes and interact with targets inside the cell.
What is the difference between binding affinity and specificity?
Binding affinity measures how tightly a peptide binds to a receptor (how firmly the key sits in the lock). Specificity measures how many different receptors a peptide can bind to (how many locks the key can open). A peptide can have high affinity for one receptor and high specificity (it only binds that one receptor), or high affinity for multiple receptors and low specificity.
Can two different peptides bind to the same receptor?
Yes. Multiple peptides can bind to the same receptor if they have compatible shapes. The body’s own natural ligand (the original key) typically has the highest affinity, but research peptides can be designed to fit the same lock. Some bind as agonists (activating the receptor) while others bind as antagonists (blocking it).
Why does peptide sequence determine receptor binding?
Because the amino acid side chains on a peptide create a unique three-dimensional shape and chemical profile. The receptor’s binding pocket has a complementary shape and chemical profile. Only peptides whose side chains match the pocket’s requirements will fit closely enough to bind effectively. Change the amino acid sequence, and you change the shape and chemistry — potentially fitting a different lock or fitting no lock at all.
For research use only. Not for human consumption. This article is intended for informational purposes and does not constitute medical advice, dosing guidance, or therapeutic recommendations.