· For research use only. Not for human consumption.
For research use only. Not for human consumption.
You pull a peptide vial from storage, reconstitute it, and something seems off. Maybe the solution looks different than usual, or maybe your research results just are not what you expected. Knowing the peptide degradation signs to watch for can help you identify compromised materials before they waste your time and resources on unreliable experiments.
Peptide degradation is a natural process that happens over time, especially when storage conditions are less than ideal. Heat, light, moisture, contamination, and repeated freeze-thaw cycles all contribute to breaking down these delicate molecules. The challenge is that degradation often happens invisibly, with no obvious warning until it starts affecting your results.
In this article, we will walk through five observable signs that your peptide may have degraded, explain what causes each one, and give you practical tips for prevention. Knowing what to look for helps you make informed decisions about whether to continue using a sample or replace it with fresh material from Alpha Peptides.
TL;DR: The five main signs of peptide degradation are: (1) discoloration or color changes, (2) unusual smell, (3) difficulty dissolving, (4) visible clumping or aggregation, and (5) unexpected or inconsistent research results. These signs indicate chemical, physical, or biological degradation. Prevention involves proper temperature storage, light protection, moisture control, sterile handling, and minimizing freeze-thaw cycles.
For research use only. Not for human consumption.
Sign 1: Changed Appearance or Discoloration: Peptide degradation signs Insights
Most research peptide solutions should be clear and colorless after reconstitution. If your solution has turned yellow, brown, or any other color, something has changed chemically. This is one of the most straightforward peptide degradation signs because you can spot it instantly.
Discoloration typically results from oxidation reactions. Certain amino acids, particularly tryptophan and methionine, are prone to oxidation when exposed to air, light, or trace metals. When these amino acids oxidize, they form colored byproducts that tint the solution.
A lyophilized powder that was originally white or off-white but has turned yellow or tan may also have degraded. This can happen if moisture entered the vial during storage, reactivating degradation reactions in what should have been a dry, stable powder.
The practical takeaway: if it does not look right, do not trust it. Discolored peptides may still contain some functional material, but the degree of degradation is impossible to assess visually. You cannot know whether 10% or 90% of the peptide has been damaged just by looking at the color.
Sign 2: Unusual Smell
Fresh peptide solutions are generally odorless or have only a very faint chemical smell. If you notice an unusual, strong, or unpleasant odor when you open a vial, it is a warning sign worth paying attention to.
A sour or foul smell often indicates bacterial contamination. Bacteria that have colonized a peptide solution produce metabolic byproducts that generate distinctive odors. If your solution smells noticeably off, contamination is the most likely explanation, and the solution should be discarded.
A strong chemical or sulfurous smell can indicate chemical degradation, particularly the breakdown of sulfur-containing amino acids like methionine and cysteine. These amino acids can produce volatile sulfur compounds when they degrade, creating noticeable odors.
If your solvent is bacteriostatic water, note that the benzyl alcohol preservative has a faint, mildly sweet smell that is normal. This should not be confused with the off-putting odors associated with degradation or contamination.
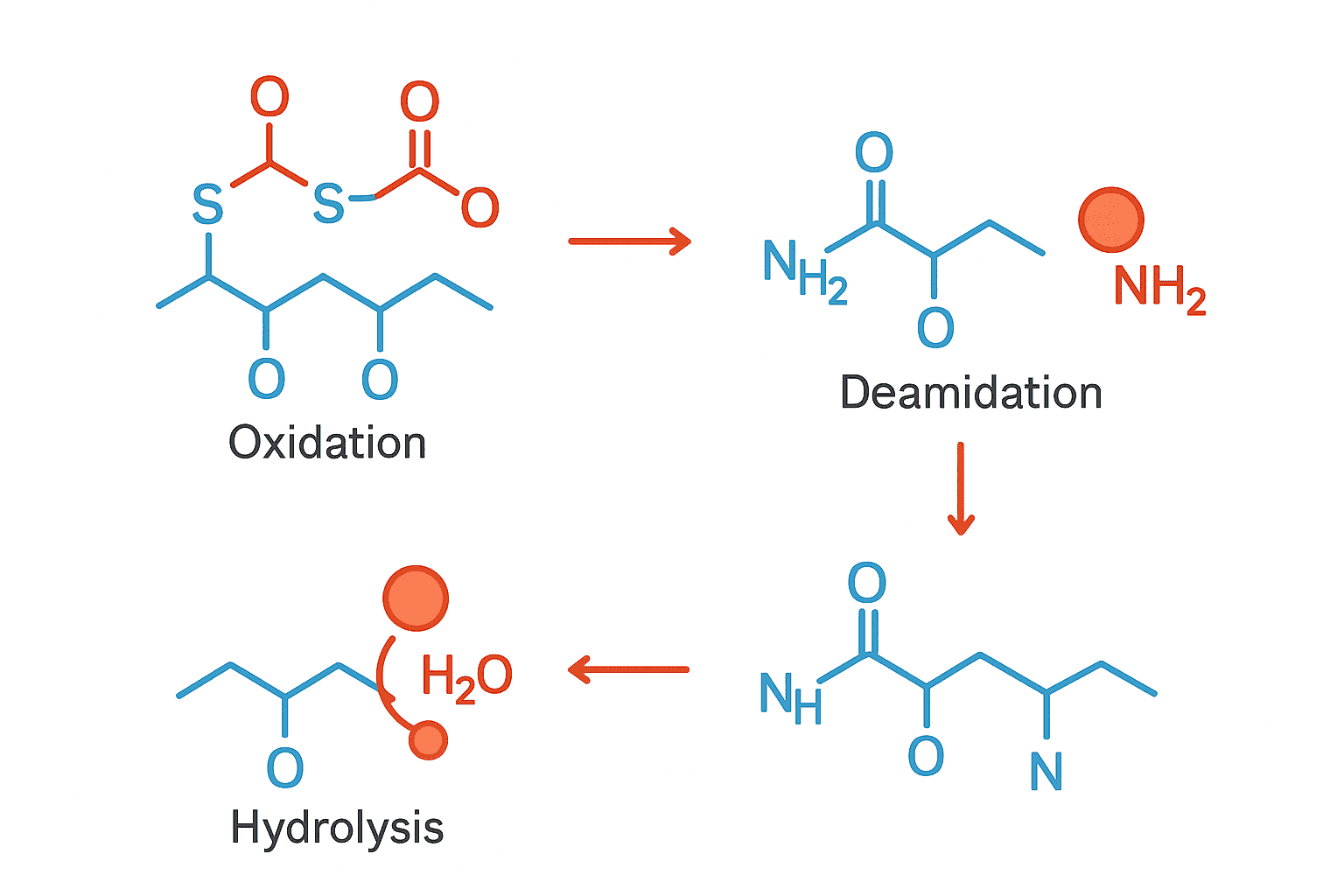
Sign 3: Difficulty Dissolving
A peptide that previously dissolved quickly and cleanly in your chosen solvent but now resists going into solution may have degraded. Degradation can change a peptide’s chemical properties, including its solubility, making it harder to dissolve than the intact molecule.
This sign is particularly relevant for lyophilized peptides. If moisture entered the vial during storage, it can cause partial degradation and aggregation of the dried powder. The resulting material may form sticky clumps or a gel-like substance that does not dissolve properly even in solvent that worked perfectly before.
That said, some peptides are naturally difficult to dissolve, so this sign is most meaningful when you notice a change from previous behavior. If a peptide that always dissolved easily in water suddenly requires extended sonication or a different solvent, degradation should be suspected.
Sign 4: Clumping and Aggregation
Aggregation is one of the most common forms of peptide degradation. When peptides lose their proper structure (denature), they can stick together in clumps called aggregates. These aggregates can be visible as floating particles, cloudiness, or a gel-like film in an otherwise clear solution.
Aggregation is caused by a variety of factors: repeated freeze-thaw cycles, exposure to extreme temperatures, vigorous shaking, and high concentrations can all promote clumping. Once aggregated, peptides are generally not recoverable — you cannot reverse the process by gentle mixing or sonication.
Small amounts of aggregation might be invisible to the naked eye but can still affect research results. If you see any visible particles, cloudiness, or gel formation in a reconstituted peptide solution, the sample has experienced significant aggregation and should be replaced.

Sign 5: Unexpected Research Results
This is the most frustrating sign because it only becomes apparent after you have already used the peptide in an experiment. If your research results suddenly become inconsistent, show reduced activity, or deviate from established patterns without any change in your protocol, degraded peptide is a prime suspect.
Degraded peptides may still partially function, which makes this sign tricky to interpret. You might see a gradual decline in activity over time as the peptide progressively breaks down, rather than a sudden and complete loss of function. This gradual decline can be mistaken for other experimental variables.
If you suspect peptide degradation is affecting your results, the most reliable test is to run your experiment with a fresh, unopened vial and compare. If the fresh vial produces the expected results and your older sample does not, degradation is the most likely explanation.
What Causes Degradation and How to Prevent It
All five signs above result from a handful of common causes. Here is a quick reference for each cause and its prevention:
- Heat: Store lyophilized peptides at -20°C or below. Keep reconstituted solutions refrigerated or frozen. Minimize time at room temperature.
- Light: Use amber vials or wrap in aluminum foil. Store in dark locations. Avoid leaving peptides under fluorescent lights.
- Moisture: Keep lyophilized vials sealed tightly. Store with desiccant packets. Do not open vials in humid environments.
- Contamination: Use proper sterile technique. Swab vial tops with alcohol. Use fresh needles. Choose bacteriostatic water for reconstitution.
- Freeze-thaw cycles: Aliquot reconstituted peptides into single-use portions before freezing. Thaw only what you need.
Prevention is always easier than detection. Starting with high-purity, properly handled peptides gives you the best foundation. Alpha Peptides provides third-party tested research peptides with batch-specific Certificates of Analysis. Browse the full catalog at alpha-peptides.com/shop.

Frequently Asked Questions
Can I still use a peptide that shows minor signs of degradation?
It depends on your research requirements. Minor signs may indicate partial degradation, meaning some functional peptide remains. However, you cannot accurately quantify the remaining activity without analytical testing. For critical experiments, replacing the sample with fresh material is the safest approach.
How can I confirm whether my peptide has actually degraded?
Analytical testing is the gold standard. HPLC (high-performance liquid chromatography) can detect degradation products and quantify remaining purity. Mass spectrometry can identify specific chemical changes. Comparing against the original Certificate of Analysis provides a reliable baseline.
Does higher purity peptide degrade more slowly?
Not necessarily. Degradation rate depends on the peptide’s amino acid sequence, storage conditions, and handling — not the initial purity. However, starting with higher purity means you have more functional peptide to begin with, giving you a larger margin before degradation affects your results.
Should I discard a peptide if only one of these signs is present?
Any single sign warrants caution. Discoloration, odor, or visible aggregation are strong indicators that the peptide should be replaced. Difficulty dissolving and inconsistent results may have other explanations but should prompt investigation. When in doubt, use a fresh sample for important experiments.
For research use only. Not for human consumption. This article is intended for informational purposes and does not constitute medical advice, dosing guidance, or therapeutic recommendations.