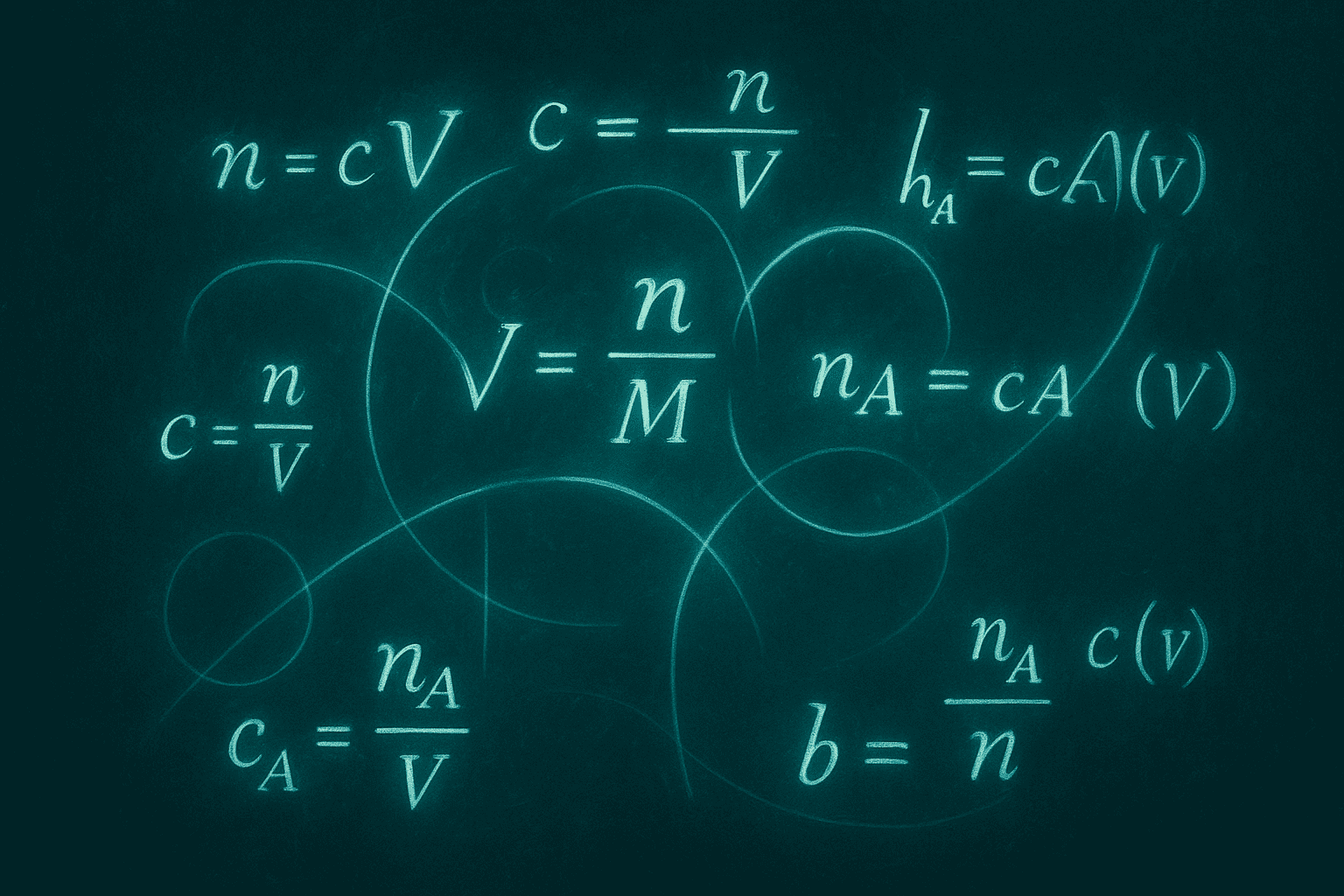· For research use only. Not for human consumption.
For research use only. Not for human consumption.
One of the first hurdles new researchers face is figuring out the math behind preparing peptide solutions. Peptide reconstitution math sounds intimidating, but it is actually just basic division and multiplication. Once you understand the simple formula, you will be able to calculate concentrations for any peptide in seconds.
The goal of reconstitution is to dissolve a known amount of freeze-dried peptide powder into a known volume of solvent, creating a solution with a precise concentration. Getting this right is essential for accurate, reproducible research. Getting it wrong means your experiments are built on inaccurate numbers from the start.
In this guide, we will walk through the math step by step using simple, real-world examples. No advanced chemistry degree required. If you can divide one number by another, you can master peptide reconstitution.
TL;DR: Peptide reconstitution math comes down to one simple formula: milligrams of peptide divided by milliliters of solvent equals concentration in mg/mL. For example, 10 mg of peptide dissolved in 2 mL of bacteriostatic water gives you a concentration of 5 mg/mL. More solvent means a more dilute solution. Less solvent means a more concentrated solution. Always record your concentration for accurate research.
For research use only. Not for human consumption.
The Basic Formula: mg ÷ mL = mg/mL: Peptide reconstitution math Insights
The entire foundation of peptide reconstitution math rests on one formula:
Concentration (mg/mL) = Amount of peptide (mg) ÷ Volume of solvent (mL)
That is it. If you know how much peptide you have (printed on the vial label in milligrams) and you decide how much solvent to add (in milliliters), you can calculate your concentration instantly.
Let us start with the simplest possible example. You have a vial containing 10 mg of peptide. You add 1 mL of bacteriostatic water. Your concentration is:
10 mg ÷ 1 mL = 10 mg/mL
Every 1 mL of your solution now contains 10 mg of peptide. That is all there is to the core calculation.
Working Through Real Examples
Let us try a few more examples to build your confidence:
Example 1: You have a 5 mg vial of peptide and add 1 mL of water.
5 mg ÷ 1 mL = 5 mg/mL
Example 2: You have a 10 mg vial of peptide and add 2 mL of water.
10 mg ÷ 2 mL = 5 mg/mL
Example 3: You have a 5 mg vial of peptide and add 2.5 mL of water.
5 mg ÷ 2.5 mL = 2 mg/mL
Example 4: You have a 15 mg vial of peptide and add 3 mL of water.
15 mg ÷ 3 mL = 5 mg/mL
Notice the pattern: adding more solvent gives you a lower concentration (more dilute), and adding less solvent gives you a higher concentration (more concentrated). You control the concentration by choosing how much solvent to add.
Why Concentration Matters for Research
You might wonder why getting the exact concentration right is so important. The answer comes down to reproducibility and accuracy — the two pillars of good research.
If you are running an experiment that requires a specific amount of peptide, you need to know exactly how much peptide is in each unit of volume you draw from your vial. Without an accurate concentration, you cannot control how much peptide goes into your experiment, and your results become unreliable.
Imagine trying to follow a recipe that says “add some flour.” Without knowing the amount, you get a different result every time. Concentration is the measurement that transforms “some peptide” into a precise, repeatable quantity. Recording your concentration alongside your experimental data allows you and other researchers to replicate the work exactly.
Working Backwards: How Much Solvent to Add
Sometimes you want a specific concentration and need to figure out how much solvent to add. The formula rearranges easily:
Volume of solvent (mL) = Amount of peptide (mg) ÷ Desired concentration (mg/mL)
Example: You have 10 mg of peptide and want a concentration of 2 mg/mL.
10 mg ÷ 2 mg/mL = 5 mL of solvent
So you would add 5 mL of bacteriostatic water to your 10 mg vial to achieve a 2 mg/mL solution.
Another example: You have 5 mg of peptide and want a concentration of 1 mg/mL.
5 mg ÷ 1 mg/mL = 5 mL of solvent
And one more: You have 30 mg and want 10 mg/mL.
30 mg ÷ 10 mg/mL = 3 mL of solvent

A Quick Note on Micrograms and Molarity
So far we have worked entirely in milligrams (mg) and milliliters (mL), which is the most common and practical way to express peptide concentrations. However, you may occasionally encounter two other units:
Micrograms (mcg or μg): One milligram equals 1,000 micrograms. Some research protocols express amounts in micrograms because they work with very small quantities. To convert, simply multiply mg by 1,000. So a 2 mg/mL solution is also a 2,000 mcg/mL solution.
Molarity (M or mM or μM): This is a chemistry-specific way of expressing concentration based on the molecular weight of the peptide rather than its mass. To calculate molarity, you divide the concentration in mg/mL by the peptide’s molecular weight (in grams per mole), then multiply by 1,000 to get millimolar (mM). You only need this for protocols that specify molar concentrations, and the molecular weight is usually listed on the certificate of analysis.
For most practical purposes, mg/mL is all you need. Only convert to other units if your specific research protocol requires it.
Practical Tips for Accurate Reconstitution
Getting the math right is only half the battle. Here are some tips to make sure your actual solution matches your calculated concentration:
- Add solvent slowly: When adding bacteriostatic water to a peptide vial, direct the stream down the inside wall of the vial rather than directly onto the powder. This prevents splashing and loss of material.
- Swirl gently, do not shake: After adding solvent, swirl the vial gently to dissolve the peptide. Vigorous shaking can create foam and promote degradation.
- Let it dissolve fully: Some peptides take a few minutes to dissolve completely. Be patient. If undissolved powder remains after five minutes of gentle swirling, the peptide may need a different solvent or different conditions.
- Label your vial: Write the concentration, date of reconstitution, and solvent used directly on the vial or on a label. This prevents confusion later.
- Use precise measurement tools: Use calibrated syringes to measure your solvent volume. Eyeballing the amount defeats the purpose of doing the math.
Start with quality materials for the best results. Alpha Peptides offers pharmaceutical-grade Hospira bacteriostatic water and high-purity research peptides with batch-specific Certificates of Analysis.

Frequently Asked Questions
Does the type of solvent affect the concentration calculation?
No. The math is the same regardless of whether you use bacteriostatic water, sterile water, DMSO, or any other solvent. Milligrams of peptide divided by milliliters of solvent always gives you mg/mL.
What if the peptide does not fully dissolve?
If undissolved material remains, your actual concentration will be lower than your calculated value because some peptide is not in solution. This can indicate a solubility issue, and you may need to try a different solvent or consult solubility guidelines for that specific peptide.
Can I add more solvent later to dilute my solution?
Yes. If your solution is too concentrated, you can add additional solvent to dilute it. Recalculate your new concentration using the total amount of peptide divided by the new total volume of solvent.
Why do some protocols list amounts in “units” instead of milligrams?
Some biological molecules have activity measured in standardized units rather than by mass. For most research peptides, mass-based concentrations (mg/mL) are the standard. If your protocol uses units, refer to the specific conversion factor provided by the peptide manufacturer or in the relevant research literature.
For research use only. Not for human consumption. This article is intended for informational purposes and does not constitute medical advice, dosing guidance, or therapeutic recommendations.