· For research use only. Not for human consumption.
For research use only. Not for human consumption.
Selank tuftsin is a pairing you’ll see often in peptide research literature, and for good reason. Selank wasn’t invented from scratch in a lab. It was built on top of tuftsin, a small peptide that your body already makes naturally. Russian scientists took that natural molecule, added three extra amino acids, and created something new. Understanding that connection helps explain why Selank became a focus of preclinical neuroscience research.
Think of it like cooking. Tuftsin is the original recipe that nature came up with. Selank is what happened when scientists took that recipe and added a few extra ingredients to make it last longer and do more in the lab. The foundation is natural. The modification is synthetic. That blend of natural origin and scientific engineering is what makes the Selank tuftsin story worth knowing about.
This guide breaks down what tuftsin is, how it was discovered, why Russian researchers chose it as a starting point, and what they changed to create Selank. For broader context on Selank’s place in neuroscience, see our Selank nootropic research overview. For comparisons with related peptides, check out our look at brain peptide research in 2026.
[INTERNAL-LINK: “Selank nootropic research overview” -> /blog/selank-nootropic-research-overview/]
[INTERNAL-LINK: “brain peptide research in 2026” -> /blog/brain-peptide-research-2026/]
TL;DR: Selank is a synthetic heptapeptide built from tuftsin, a naturally occurring four-amino-acid immune peptide discovered in the 1970s. Russian scientists at the Institute of Molecular Genetics extended tuftsin by adding a Pro-Gly-Pro tail to create a more stable research compound. Seredenin et al. (1998) documented the anxiolytic action of this tuftsin analog in preclinical models (PMID: 9583175). Selank is sold for research use only.
What Is Tuftsin and Why Does It Matter?
Tuftsin is a peptide made of just four amino acids: threonine, lysine, proline, and arginine. Written in shorthand, that’s Thr-Lys-Pro-Arg. Your body produces it naturally. It comes from a larger protein called leukokinin, which is made in the spleen. When leukokinin gets broken down by enzymes, tuftsin is one of the fragments that gets released.
The peptide was first identified in the early 1970s by a scientist named Victor Najjar at Tufts University in Boston. He named it “tuftsin” after his institution. His research found that this tiny fragment played a role in how certain immune cells behaved — specifically, it was connected to phagocytosis, the process where immune cells engulf and break down unwanted material.
What made tuftsin interesting to scientists wasn’t just what it did but how small it was. Four amino acids is remarkably short for a biologically active molecule. That simplicity made it an attractive starting point for researchers who wanted to build something new on top of a proven natural foundation.
Seredenin et al. (1998) examined the anxiolytic action of a tuftsin analog (Selank) in preclinical behavioral models. The study established that this modified version of tuftsin displayed properties distinct from its parent molecule, opening a new direction for tuftsin-derived peptide research. (PMID: 9583175)
How Russian Scientists Modified Tuftsin to Create Selank
In the 1990s, researchers at the Institute of Molecular Genetics in Moscow saw potential in tuftsin’s compact structure. They wanted to explore whether extending it could produce a compound with different properties worth investigating. Their approach was straightforward: take tuftsin’s four amino acids and attach three more to the end.
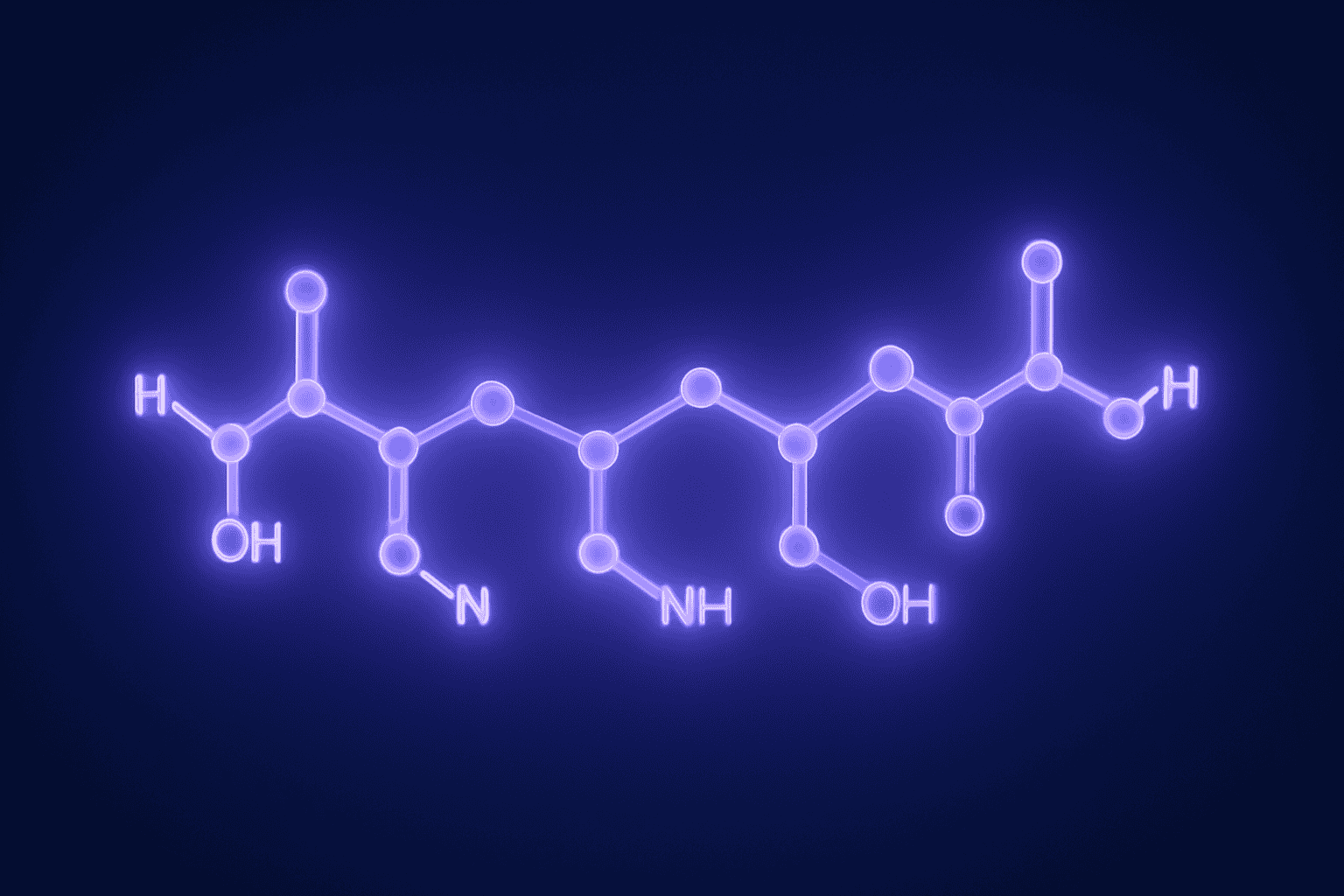
The three amino acids they added were proline, glycine, and proline again — abbreviated as Pro-Gly-Pro. This extension wasn’t random. The Pro-Gly-Pro tail acts like a protective cap. Without it, enzymes in biological environments would break down tuftsin in minutes. With it, the resulting seven-amino-acid chain resists enzymatic degradation much longer, making it far more practical for laboratory investigation.
The final product — Thr-Lys-Pro-Arg-Pro-Gly-Pro — is what we now call Selank. Seven amino acids total. Four from nature’s original recipe, three added by scientists. The Selank tuftsin relationship is a textbook example of how researchers take something the body already produces and adapt it for preclinical study.
Why Starting From a Natural Peptide Matters for Selank Tuftsin Research

When researchers design synthetic peptides, they have two basic choices. They can create something completely new from scratch, or they can start from a molecule that already exists in biology and modify it. The Selank tuftsin approach falls into the second category, and that choice has real consequences for research.
Starting from tuftsin meant that the core structure of Selank already had a known biological context. Tuftsin had been studied for decades before Selank was ever created. Scientists knew which enzymes broke it down, what cell receptors interacted with it, and how it behaved in different environments. That background knowledge gave Selank researchers a head start.
Kozlovskaya et al. (2003) investigated Selank alongside other short peptides of the tuftsin family, examining how structural modifications to the tuftsin core changed the compound’s research profile (PMID: 14969422). Their work demonstrated that small changes to a natural peptide can produce meaningfully different results in preclinical settings. The tuftsin backbone provides the foundation; the Pro-Gly-Pro extension changes what that foundation can do.
Kozlovskaya et al. (2003) studied Selank and short peptides of the tuftsin family, characterizing how modifications to the tuftsin sequence affected preclinical research outcomes. This work placed Selank within a broader family of tuftsin-derived compounds and contributed to understanding structure-activity relationships in short peptides. (PMID: 14969422)
What Has Preclinical Research Found About Selank?
Since Selank was developed, it has appeared in a number of preclinical studies examining different aspects of its behavior. Seredenin et al. (1998) were among the first to document anxiolytic-like activity of this tuftsin analog in animal models (PMID: 9583175). The animals displayed reduced anxiety-type behavior in standard behavioral tests without showing signs of sedation.
Later studies expanded into other areas. Kolik et al. (2019) investigated Selank in the context of ethanol-induced memory impairment, finding that it was connected to regulation of BDNF (brain-derived neurotrophic factor) expression in preclinical models (PMID: 31625062). BDNF is a protein involved in how neurons grow and form connections, so this finding opened another research direction for the Selank tuftsin compound.
It’s worth noting that all of this research remains at the preclinical stage. These are observations from animal models and in vitro studies, not clinical trials with people. The data is published and peer-reviewed, but it represents early-stage scientific investigation.
The Selank Tuftsin Connection: A Simple Analogy

If chemistry analogies make your eyes glaze over, try this one instead. Imagine tuftsin as a four-letter word. It makes sense on its own and has its own meaning in biology. Now imagine scientists adding three more letters to that word, creating a seven-letter word with a related but different meaning. The original word is still in there — you can still see it — but the new word does something the original couldn’t.
That’s the Selank tuftsin connection in a nutshell. The first four amino acids are tuftsin’s original sequence, and they remain unchanged. The last three amino acids are the scientific modification that transformed a fragile immune fragment into a more stable research compound. Same root, new capabilities for laboratory investigation.
For researchers interested in exploring Selank’s properties or comparing it with other brain-related peptides, our overview of Semax and its ACTH origins provides a useful parallel case of how scientists build on natural peptide fragments.
[INTERNAL-LINK: “Semax and its ACTH origins” -> /blog/semax-acth-fragment-explained/]
Where Can Researchers Source Selank?
Research-grade Selank requires verified purity documentation. Look for a supplier providing third-party HPLC purity data (minimum 98%), mass spectrometry confirmation of the correct molecular weight, and batch-specific Certificates of Analysis from an independent laboratory.
Alpha Peptides carries research-grade Selank with publicly available COAs. You can review documentation on our Certificates of Analysis page or browse the full research catalog.
[INTERNAL-LINK: “Certificates of Analysis page” -> /coas/]
[INTERNAL-LINK: “research catalog” -> /shop/]
Frequently Asked Questions
What is tuftsin?
Tuftsin is a naturally occurring four-amino-acid peptide (Thr-Lys-Pro-Arg) produced by the spleen. It was discovered in the 1970s at Tufts University and is connected to immune cell phagocytosis. It serves as the structural foundation for Selank.
How is Selank related to tuftsin?
Selank is a synthetic extension of tuftsin. Russian scientists added three amino acids (Pro-Gly-Pro) to tuftsin’s four-amino-acid chain, creating a seven-amino-acid compound. The Selank tuftsin relationship is one of parent molecule to modified analog. Seredenin et al. (1998) first characterized this tuftsin analog’s properties in preclinical research (PMID: 9583175).
Why did scientists add Pro-Gly-Pro to tuftsin?
The Pro-Gly-Pro tail acts as an enzymatic shield. Without it, tuftsin would break down very quickly in biological environments. Adding this three-amino-acid extension made the resulting compound (Selank) more resistant to degradation, which is essential for practical laboratory research.
Is Selank the same as tuftsin?
No. While Selank contains tuftsin’s full amino acid sequence as its core, the addition of Pro-Gly-Pro makes it a distinct compound with different stability characteristics and research applications. Think of tuftsin as the foundation and Selank as the finished structure built on top of it.

For research use only. Not for human consumption. All peptides referenced in this article are intended exclusively for laboratory and preclinical research purposes. Nothing on this page constitutes medical advice, dosing guidance, or a recommendation for personal use. All information is provided for educational purposes relating to peptide chemistry and laboratory research practice.