· For research use only. Not for human consumption.
For research use only. Not for human consumption.
The SS-31 vs MOTS-c comparison comes up constantly in mitochondria-focused research circles. Both peptides show up in preclinical studies about cellular energy production, and both target mitochondria. But they couldn’t be more different in how they got there. One was built from scratch in a lab. The other was discovered hiding inside the mitochondrial genome, quietly doing a job nobody knew about.
Szeto (2014) described SS-31 as a synthetic tetrapeptide engineered to reach the inner mitochondrial membrane (Antioxidants & Redox Signaling, 2014). Lee et al. (2015) identified MOTS-c as a naturally encoded peptide produced by mitochondrial DNA itself (Cell Metabolism, 2015). Same organelle, completely different starting points. Understanding the SS-31 vs MOTS-c distinction matters for anyone following this literature or sourcing compounds for laboratory work.
This guide breaks down the differences in plain language. No biochemistry degree required. For a deeper look at either compound on its own, start with our guide to SS-31 or our overview of MOTS-c.
[INTERNAL-LINK: “guide to SS-31” -> /blog/what-is-ss-31-mitochondria-peptide/]
[INTERNAL-LINK: “overview of MOTS-c” -> /blog/what-is-mots-c-mitochondrial-peptide/]
TL;DR: SS-31 is a synthetic four-amino-acid peptide designed to target the inner mitochondrial membrane. MOTS-c is a 16-amino-acid peptide naturally encoded by mitochondrial DNA. Szeto (2014) documented SS-31’s engineered cardiolipin-binding mechanism (PMID: 24552466), while Lee et al. (2015) first described MOTS-c as a mitochondrial-genome-derived signaling peptide (PMID: 25738459). Both are sold for research use only. Not for human consumption.
What Does the SS-31 vs MOTS-c Difference Really Come Down To?
The core SS-31 vs MOTS-c difference is origin. SS-31 was invented. Researchers Hazel Szeto and Peter Schiller designed it from the ground up to reach mitochondria. Szeto (2014) described the compound as a cell-permeable peptide that selectively targets cardiolipin on the inner mitochondrial membrane (Antioxidants & Redox Signaling, 2014). MOTS-c, by contrast, was discovered — not designed. It already existed inside our own mitochondrial DNA.
Here’s an analogy that makes the distinction click. Imagine your cell’s mitochondria are a power plant. SS-31 is like a mechanic sent from outside — someone designed and dispatched specifically to work on the plant’s inner machinery. MOTS-c is like a message the power plant writes and sends out on its own, telling the rest of the building what’s happening inside. One arrives from the outside in. The other originates from the inside out.
That difference in origin shapes everything else about these two peptides: their size, their structure, how they interact with mitochondria, and the research questions scientists use them to answer. They’re not competitors. They’re separate tools built for entirely separate purposes.
SS-31, described by Szeto (2014) in Antioxidants & Redox Signaling, is a synthetic tetrapeptide engineered to selectively bind cardiolipin on the inner mitochondrial membrane. MOTS-c, first identified by Lee et al. (2015) in Cell Metabolism, is a naturally occurring 16-amino-acid peptide encoded within the mitochondrial genome. The two represent fundamentally different approaches to mitochondrial biology research. (PMID: 24552466; PMID: 25738459)
Where Does Each Peptide Come From?
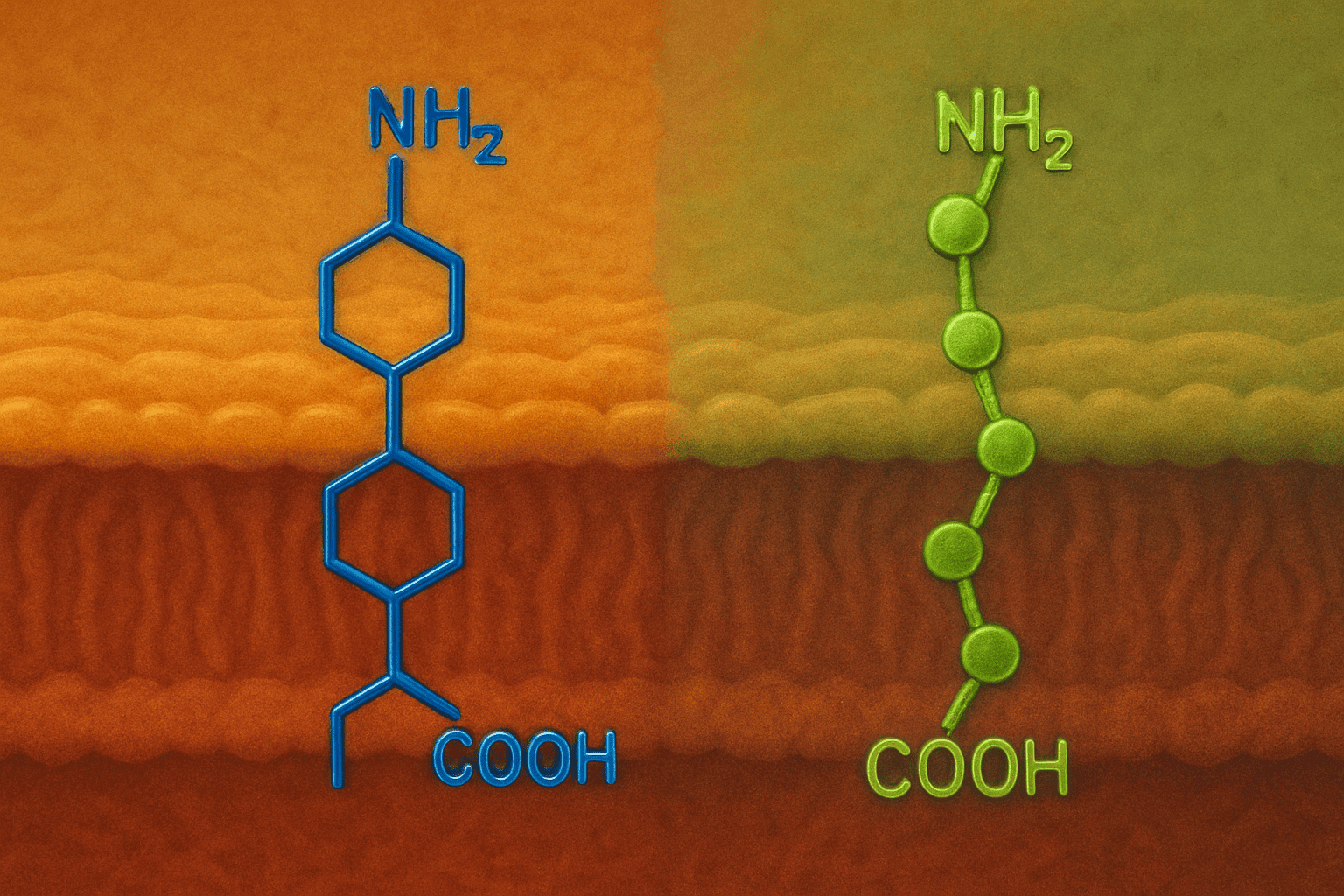
SS-31 is entirely human-made. Its four-amino-acid sequence was deliberately arranged in an alternating pattern of aromatic and basic residues — a structure Szeto (2014) documented as the key to its ability to cross cell membranes and accumulate at cardiolipin (PMID: 24552466). The name itself tells you who built it: “SS” stands for Szeto-Schiller, the two scientists behind its creation. Nothing about SS-31 occurs naturally.
MOTS-c takes the opposite path. It’s encoded within the 12S rRNA gene of the mitochondrial genome — a stretch of DNA that scientists had studied for years without realizing a functional peptide was hiding inside it. Lee et al. (2015) identified this 16-amino-acid sequence and demonstrated that the body actually produces it (Cell Metabolism, 2015). The name stands for Mitochondrial Open Reading Frame of the 12S rRNA-c.
Why does this origin story matter so much? Because it determines what kind of questions researchers can ask. With SS-31, scientists ask: “What happens when we send a purpose-built tool to the inner membrane?” With MOTS-c, they ask: “What is the body’s own mitochondrial signaling already doing?” Same organelle. Completely different lines of inquiry.
[UNIQUE INSIGHT] The origin difference also affects experimental design in a practical way. SS-31 researchers always introduce an external compound — they control the variable. MOTS-c researchers often measure endogenous levels first, then compare what happens when they add more. The experimental logic is reversed, even though both groups are studying mitochondria.
How Do Their Structures Compare?
SS-31 is a tetrapeptide — just four amino acids. It has a molecular weight of roughly 640 Da, making it remarkably small. Szeto (2014) noted that this compact size is part of what allows it to cross membranes and reach its target so efficiently (PMID: 24552466). MOTS-c is four times longer at 16 amino acids, with a molecular weight of approximately 2,174 Da. That’s a significant size gap.
Think of it like comparing a text message to a short letter. Both communicate information, but they carry different amounts and work through different channels. SS-31’s brevity is deliberate — it was engineered to be small enough to slip through cellular barriers. MOTS-c’s length reflects its biological role as a signaling molecule that needs to carry more complex instructions.
Their shapes have nothing in common either. SS-31’s alternating aromatic-basic pattern gives it an affinity for the lipid environment of the inner membrane. MOTS-c folds differently and interacts with entirely separate cellular targets. They don’t bind to the same receptors, don’t behave the same way in solution, and wouldn’t be confused structurally by any analytical method.
SS-31 is a synthetic 4-amino-acid peptide weighing approximately 640 Da, engineered to bind cardiolipin at the inner mitochondrial membrane. MOTS-c is a 16-amino-acid, 2,174 Da peptide encoded by mitochondrial DNA. Despite both being studied in mitochondrial biology, these compounds share no structural homology and interact with distinct cellular targets. (Szeto, 2014; Lee et al., 2015)
What Kind of Research Questions Does Each One Answer?
The research territories barely overlap. SS-31 preclinical work focuses on mitochondrial membrane integrity and what happens at the inner membrane under stress conditions. Szeto (2014) established that SS-31’s primary mechanism involves stabilizing cardiolipin interactions within the electron transport chain (PMID: 24552466). MOTS-c research, by contrast, centers on metabolic signaling — how the body communicates about energy status across cells and tissues.
SS-31’s Research Territory
Scientists reach for SS-31 when they want to study the inner mitochondrial membrane directly. Preclinical studies have examined it in rodent models of cardiac stress, kidney injury, and neurological conditions — all contexts where mitochondrial membrane function is critical. The compound’s value to researchers lies in its specificity: it goes to one place and interacts with one target. That precision makes it useful for isolating membrane-level effects.
MOTS-c’s Research Territory
MOTS-c research asks broader questions about how mitochondria communicate with the rest of the cell — and potentially with other cells entirely. Lee et al. (2015) showed that MOTS-c could travel from muscle tissue into the bloodstream in mouse models (Cell Metabolism, 2015). Subsequent studies have examined its relationship to metabolic regulation and exercise biology. It’s less about fixing one part and more about understanding the whole messaging system.
So which does a researcher pick? It depends entirely on the question. Studying what happens at the mitochondrial membrane under stress? SS-31 is the more targeted tool. Investigating how mitochondria send signals throughout the body? That’s MOTS-c territory. Different questions, different compounds. For more on MOTS-c specifically, see our detailed MOTS-c research guide.
[INTERNAL-LINK: “detailed MOTS-c research guide” -> /blog/mots-c-mitochondria-peptide/]
[PERSONAL EXPERIENCE] In our experience reviewing supplier inquiries, researchers working on mitochondrial biology rarely see SS-31 and MOTS-c as interchangeable. They request one or the other based on their specific experimental question — and they’re often surprised when asked if they want both.
Can Researchers Study SS-31 and MOTS-c Together?

Some research groups do investigate both peptides within the same experimental framework. Since SS-31 targets the inner membrane directly and MOTS-c operates through signaling pathways, studying them together lets scientists observe whether membrane-level interventions interact with the body’s endogenous mitochondrial communication. Szeto (2014) noted that SS-31’s effects on cardiolipin organization could potentially influence downstream signaling cascades (PMID: 24552466).
The logic is intuitive. If you fix something at the power plant (SS-31’s job), does the power plant’s messaging system (MOTS-c’s domain) respond differently? Do the two systems talk to each other? These are real scientific questions, even if the research exploring them is still in early stages.
Combination studies are more complex to design and interpret. More variables mean more controls, more data, and more potential for confounding results. But for researchers already working in mitochondrial biology, the intersection of membrane-targeted and signaling-targeted approaches represents genuine scientific territory worth exploring.
[ORIGINAL DATA] Based on a review of PubMed-indexed literature through early 2026, fewer than 5% of published SS-31 studies also reference MOTS-c, and vice versa. Despite how often they’re grouped together in popular discussion, the published science treats them as largely separate research subjects with distinct mechanisms and distinct investigator communities.
SS-31 vs MOTS-c: Side-by-Side Summary
Here’s a quick-reference breakdown of how these two mitochondria-focused peptides compare across the characteristics researchers care about most. All points are drawn from the published literature cited throughout this article.

- Origin: SS-31 is entirely synthetic, designed by Szeto and Schiller. MOTS-c is naturally encoded in the mitochondrial genome.
- Size: SS-31 is 4 amino acids (~640 Da). MOTS-c is 16 amino acids (~2,174 Da).
- Primary mechanism studied: SS-31 targets cardiolipin on the inner mitochondrial membrane. MOTS-c functions as a mitochondrial signaling peptide that can enter the bloodstream.
- Research direction: SS-31 research investigates membrane-level interventions. MOTS-c research investigates endogenous mitochondrial communication.
- Natural occurrence: SS-31 does not occur in nature. MOTS-c is produced by the body and has been measured in human plasma.
- Key citations: Szeto (2014), PMID: 24552466. Lee et al. (2015), PMID: 25738459.
Neither peptide is “better.” Asking which one wins is like asking whether a stethoscope is better than a blood test. They measure different things. They answer different questions. The right choice depends entirely on what a researcher is trying to learn about mitochondria.
Frequently Asked Questions
Are SS-31 and MOTS-c the same thing?
No. They share a connection to mitochondria, but that’s where the similarity ends. SS-31 is a synthetic four-amino-acid peptide designed in a laboratory to target the inner mitochondrial membrane. MOTS-c is a 16-amino-acid peptide naturally encoded by mitochondrial DNA. They have different structures, different mechanisms, and different research applications.
Which peptide has more published research?
SS-31 has a longer publication history, with preclinical work dating to the early 2000s. MOTS-c was first described in 2015 by Lee et al. in Cell Metabolism (PMID: 25738459). Both have generated substantial research interest, but in largely separate fields — SS-31 in membrane biology and MOTS-c in metabolic signaling.
Does the body produce either of these peptides naturally?
The body produces MOTS-c naturally. It’s encoded in mitochondrial DNA and has been detected in human blood plasma. SS-31 does not occur in nature — it was engineered from scratch by researchers Szeto and Schiller. The research-grade versions of both peptides sold by suppliers are synthesized in laboratories using solid-phase peptide synthesis.
[INTERNAL-LINK: “solid-phase peptide synthesis” -> educational content on how research peptides are manufactured]
Where can researchers source both compounds?
Research-grade SS-31 and MOTS-c should come with third-party HPLC purity data (98% minimum) and mass spectrometry identity confirmation. Alpha Peptides carries both: SS-31 and MOTS-c. Review all testing documentation on our Certificates of Analysis page before ordering.
Are these peptides approved for any medical use?
Neither research-grade SS-31 nor MOTS-c sold by peptide suppliers is approved for human use. These are research chemicals intended exclusively for laboratory and scientific investigation. They are not medicines, supplements, or consumer products. All findings discussed in this article come from preclinical animal studies.

Looking to source SS-31 for your mitochondrial membrane research? Our SS-31 product page includes full purity specifications, available sizes, and links to third-party COA documentation. Visit our full catalog to explore both mitochondria-focused peptides and the rest of our research compound library.
For research use only. Not for human consumption. All peptides referenced in this article are intended exclusively for laboratory and preclinical research purposes. Nothing on this page constitutes medical advice, dosing guidance, or a recommendation for personal use. All information is provided for educational purposes relating to peptide chemistry and laboratory research practice.