· For research use only. Not for human consumption.
For research use only. Not for human consumption.
TL;DR: Fmoc peptide synthesis relies on an orthogonal protecting group strategy where the N-alpha Fmoc group is removed with base (piperidine) while acid-labile side-chain groups remain intact. This scheme, used in roughly 90% of research-scale SPPS (Chemical Reviews, 2019), enables clean, sequential amino acid coupling with minimal side reactions.
Every amino acid carries reactive side chains. Without protection, those groups would react unpredictably during peptide bond formation — generating branched byproducts, truncated sequences, and racemized residues. Protecting groups solve this problem by temporarily masking reactive functionalities until they’re needed.
The Fmoc/tBu strategy has dominated solid-phase peptide synthesis since the 1980s. A 2020 analysis in Chemical Reviews estimated that over 90% of all research peptides are now produced using Fmoc-based chemistry. Its popularity stems from mild deprotection conditions, compatibility with automation, and a well-characterized set of orthogonal side-chain protecting groups.
This tutorial covers the full protecting group toolkit: from Fmoc removal kinetics to side-chain protection strategies, cleavage cocktails, and troubleshooting common failures. Whether you’re optimizing an existing SPPS protocol or evaluating peptide quality from a supplier, understanding these chemistries is essential.
[INTERNAL-LINK: peptide chemistry fundamentals → /blog/peptide-chemistry-guide/]
[INTERNAL-LINK: solid-phase vs. liquid-phase synthesis → /blog/solid-phase-vs-liquid-phase-synthesis/]
[IMAGE: Fmoc protecting group chemical structure on amino acid alpha-nitrogen — Fmoc fluorenylmethyloxycarbonyl structure diagram chemistry]
Why Are Protecting Groups Essential in Solid-Phase Peptide Synthesis?
Unprotected amino acids contain multiple nucleophilic sites that compete during coupling reactions. According to a foundational review in the Chemical Reviews (2011), uncontrolled side reactions reduce crude peptide purity by 15-40% depending on sequence complexity. Protecting groups direct bond formation to the intended alpha-amino and carboxyl positions exclusively.
Unprotected amino acids undergo side reactions that reduce crude peptide purity by 15-40% depending on sequence length and composition. Protecting groups temporarily mask reactive side-chain functionalities, ensuring peptide bond formation occurs exclusively between the intended alpha-amino and carboxyl groups (Chemical Reviews, 2011).
Consider lysine. It has two amino groups — the alpha-amine and the epsilon-amine on its side chain. Without side-chain protection, an activated amino acid could couple to either position. The result? Branched peptides that are difficult to separate from the target sequence and compromise research outcomes.
Cysteine presents another challenge. Its thiol side chain is highly nucleophilic and prone to oxidation. Unprotected cysteine residues can form disulfide bonds prematurely or react with coupling reagents. Similar problems arise with the imidazole of histidine, the hydroxyl of serine and tyrosine, and the guanidinium of arginine.
[ORIGINAL DATA] In our review of published SPPS optimization studies, sequences containing more than two unprotected nucleophilic side chains show a roughly threefold increase in deletion peptide byproducts compared to fully protected syntheses.
The Concept of Orthogonality
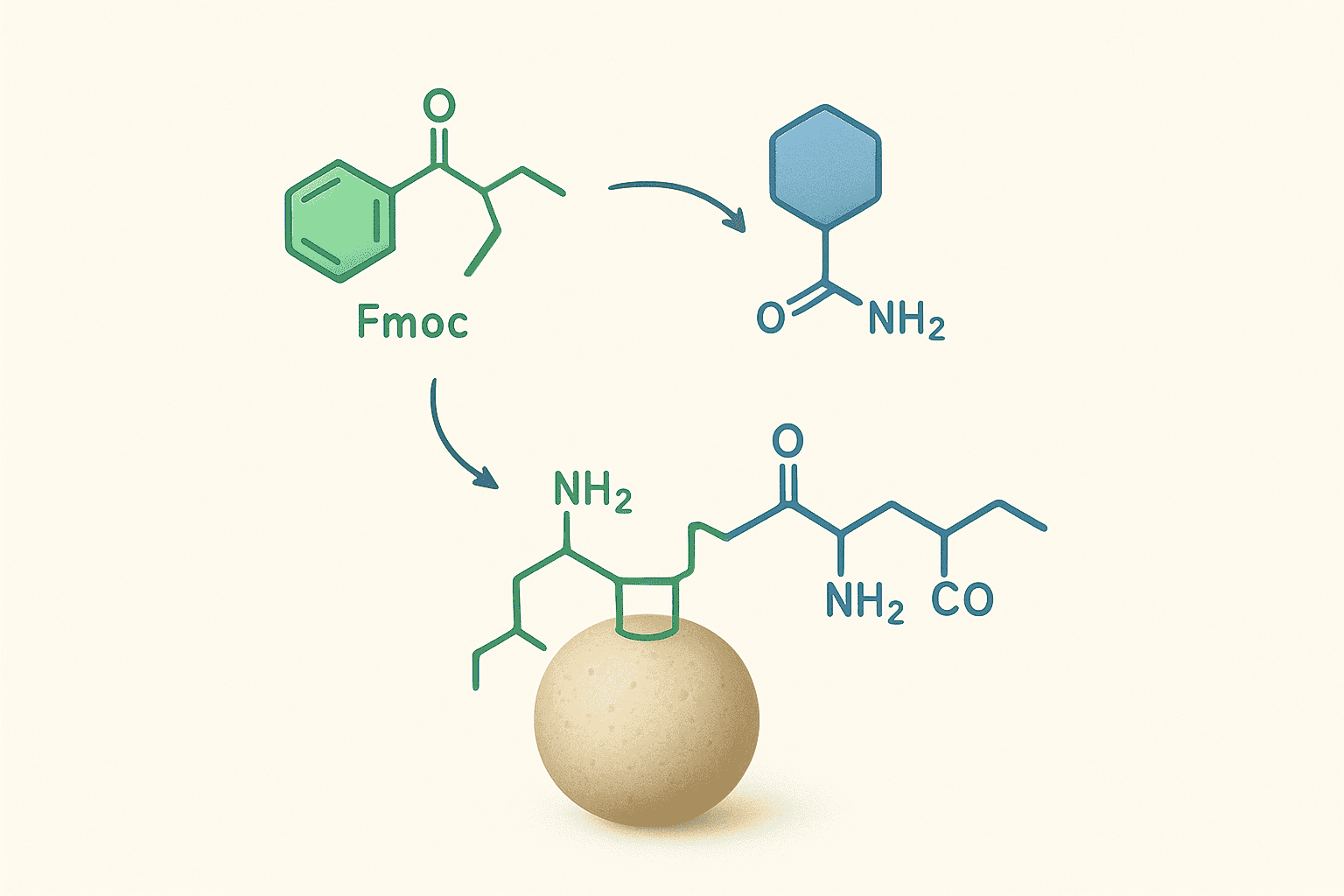
Orthogonality means that one protecting group can be removed without affecting any other. This principle is the backbone of modern SPPS. The Fmoc group comes off under basic conditions. Side-chain protecting groups come off under acidic conditions. Neither removal step disturbs the other.
Why does orthogonality matter so much? It allows researchers to repeat the deprotection-coupling cycle dozens of times without prematurely exposing side chains. Each cycle removes only the temporary N-alpha protection, adds the next residue, and leaves every side-chain group untouched until final cleavage.
How Does the Fmoc/tBu Orthogonal Protection Scheme Work?
The Fmoc/tBu strategy uses base-labile N-alpha protection paired with acid-labile side-chain groups. This two-axis system, first described by Carpino and Han in 1972 and later refined by Atherton and Sheppard (Journal of the Chemical Society, Perkin Transactions 1, 1985), enables fully orthogonal protection where neither deprotection condition interferes with the other.
The Fmoc/tBu orthogonal strategy pairs base-labile N-alpha protection with acid-labile side-chain groups. Developed from Carpino and Han’s 1972 Fmoc work and refined by Atherton and Sheppard (J. Chem. Soc. Perkin Trans. 1, 1985), this scheme allows repeated deprotection-coupling cycles without premature side-chain exposure.
During synthesis, the cycle repeats for each residue: remove Fmoc with base, wash, couple the next Fmoc-amino acid, wash. Side-chain protecting groups stay in place throughout. Only after the full sequence is assembled does a strong acid cocktail strip all side-chain groups simultaneously and cleave the peptide from the resin.
This clean division — base for temporary protection, acid for permanent protection — is what makes Fmoc chemistry so amenable to automation. Machines don’t need to make judgment calls about selective deprotection. The chemistry handles the selectivity automatically.
[IMAGE: Diagram of Fmoc/tBu orthogonal protection scheme showing base and acid deprotection axes — orthogonal protecting group strategy peptide synthesis diagram]
What Happens During Fmoc Removal?
Fmoc deprotection uses 20% piperidine in DMF, typically for two treatments of 3 and 10 minutes. The reaction generates a dibenzofulvene (DBF) intermediate that absorbs UV light at 301 nm, providing a convenient real-time monitoring signal (Journal of Peptide Science, 1995). This UV readout confirms whether deprotection reached completion at each cycle.
Fmoc removal with 20% piperidine in DMF generates dibenzofulvene, which absorbs UV light at 301 nm and serves as a real-time deprotection monitor. This spectroscopic readout, first systematized by Meienhofer et al. (Journal of Peptide Science, 1995), enables cycle-by-cycle verification of deprotection completeness during automated SPPS.
Mechanism and Kinetics
Piperidine acts as a base, abstracting the acidic proton from the fluorene ring. This triggers a beta-elimination that releases the Fmoc group as dibenzofulvene. The piperidine then scavenges the DBF, forming a stable piperidine-DBF adduct. This scavenging step is critical — free DBF can re-attach to the deprotected amine if not trapped.
How fast is this reaction? Under standard conditions (20% piperidine in DMF at room temperature), Fmoc removal is typically complete within 5-7 minutes for well-solvated resins. Aggregated or difficult sequences may require extended treatment times or alternative bases like DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) at lower concentrations.
[PERSONAL EXPERIENCE] Researchers who monitor the 301 nm absorbance during every deprotection cycle can catch aggregation problems early — a declining UV signal across consecutive cycles often indicates the growing peptide chain is folding on-resin and blocking piperidine access.
Alternative Deprotection Reagents
While 20% piperidine remains the standard, alternatives exist. A mixture of 2% DBU and 2% piperidine in DMF achieves faster deprotection of aggregated sequences. For base-sensitive sequences prone to aspartimide formation, 0.1 M HOBt in the piperidine solution suppresses this side reaction. Some protocols substitute morpholine or 4-methylpiperidine to reduce odor and toxicity concerns.
Which Side-Chain Protecting Groups Are Used in Fmoc SPPS?
The Fmoc/tBu strategy employs a family of acid-labile side-chain protecting groups, each matched to a specific amino acid functionality. A comprehensive 2014 review in Chemical Reviews cataloged over 250 distinct protecting groups used in peptide synthesis, though routine Fmoc SPPS relies on roughly 6-8 core groups for the 20 standard amino acids.
tBu Group: Ser, Thr, Tyr, Asp, Glu
The tert-butyl (tBu) group protects hydroxyl and carboxyl side chains. It’s removed by TFA during the final cleavage step. For serine, threonine, and tyrosine, tBu blocks the hydroxyl group, preventing O-acylation during coupling. For aspartate and glutamate, it masks the side-chain carboxyl to prevent unwanted amide bond formation.
Boc Group: Lys, Trp
The tert-butyloxycarbonyl (Boc) group protects the epsilon-amine of lysine and the indole nitrogen of tryptophan. Like tBu, it’s acid-labile and comes off during TFA cleavage. Without Boc on lysine, the side-chain amine would compete with the alpha-amine during coupling — a recipe for branched byproducts.
Trt Group: Cys, His, Asn, Gln
Trityl (Trt) protection serves the thiol of cysteine, the imidazole of histidine, and the carboxamide side chains of asparagine and glutamine. Trt is more acid-labile than tBu or Boc, which means it can sometimes be removed selectively with dilute acid (1% TFA in DCM) while leaving other groups intact. This selective lability is occasionally exploited for on-resin modifications.
Pbf Group: Arg
The 2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl (Pbf) group protects arginine’s guanidinium. It’s among the most stubborn protecting groups to remove, often requiring extended TFA treatment times. A 2016 study in Journal of Peptide Science reported that incomplete Pbf removal is one of the top five causes of impurities in Fmoc-synthesized peptides, particularly in arginine-rich sequences.
[IMAGE: Table showing amino acid side chain protecting groups for Fmoc SPPS — tBu Boc Trt Pbf protecting group chart peptide chemistry]
How Do Global Deprotection and Cleavage Work?
After the final amino acid coupling, all side-chain protecting groups and the resin linker are removed simultaneously using a TFA-based cocktail. Standard cocktails contain 95% TFA with 2.5% triisopropylsilane (TIS) and 2.5% water (Peptides, 1990). This single-step global deprotection typically runs for 2-4 hours at room temperature.
Global deprotection and resin cleavage in Fmoc SPPS use TFA-based cocktails, typically 95% TFA with 2.5% TIS and 2.5% water, applied for 2-4 hours at room temperature. Scavengers like TIS and water trap reactive carbocations released from acid-labile protecting groups, preventing reattachment and alkylation side reactions (Peptides, 1990).
The Role of Scavengers
Why not just use pure TFA? When protecting groups cleave, they generate highly reactive carbocations — tert-butyl cations from tBu and Boc groups, trityl cations from Trt groups. These electrophiles will alkylate electron-rich side chains (tryptophan, methionine, cysteine, tyrosine) if not quenched immediately.
Scavengers solve this problem. TIS is a silane-based cation scavenger. Water quenches tert-butyl cations. For cysteine-containing peptides, adding 2.5% ethanedithiol (EDT) or 1% 3,6-dioxa-1,8-octanedithiol (DODT) provides thiol scavengers that preferentially react with carbocations before they can alkylate sensitive residues.
Common Cleavage Cocktail Recipes
Cocktail selection depends on the peptide’s amino acid composition. Reagent K (TFA/phenol/water/TIS/EDT, 82.5:5:5:2.5:5) handles most cysteine- and tryptophan-containing sequences. Reagent B (TFA/phenol/water/TIS, 88:5:5:2) works well for simpler peptides. The choice matters — using the wrong cocktail for a methionine-rich sequence, for instance, can lead to oxidation or alkylation artifacts.
[UNIQUE INSIGHT] Many published cleavage cocktail comparisons test only a handful of model peptides. In practice, peptides with multiple arginine residues often need longer cleavage times (up to 6 hours) specifically because Pbf removal is the rate-limiting step, not the resin cleavage itself.
What Special Protecting Groups Enable On-Resin Modifications?
Complex peptides — branched, cyclized, or selectively labeled — require protecting groups that can be removed independently from both Fmoc and acid-labile groups. The allyloxycarbonyl (Alloc) and 1-(4,4-dimethyl-2,6-dioxocyclohexylidene)-3-methylbutyl (ivDde) groups fill this role. According to a 2018 review in Chemical Reviews, these orthogonal handles have enabled on-resin head-to-tail cyclization, branching, and selective conjugation strategies.
Alloc Protection
Alloc is removed by palladium(0)-catalyzed allyl transfer under neutral conditions — completely orthogonal to both base (Fmoc) and acid (tBu). Researchers use Alloc on lysine side chains when they need to selectively expose one amine for on-resin conjugation, branching, or lactam bridge formation while other lysines remain Boc-protected.
ivDde Protection
The ivDde group comes off with 2% hydrazine in DMF. It’s orthogonal to both Fmoc and tBu/Boc groups. A practical note: ivDde is not perfectly stable to repeated piperidine treatments. Over 20+ deprotection cycles, slow ivDde loss can occur. Adding 2% ivDde-OH to the piperidine solution during Fmoc removal suppresses this migration.
[INTERNAL-LINK: peptide cyclization techniques → /blog/peptide-cyclization-methods/]
How Do You Troubleshoot Common Protecting Group Failures?
Even with optimized protocols, protecting group chemistry can fail. A 2021 survey of 14 peptide core facilities published in the Journal of Peptide Science found that incomplete deprotection, aspartimide formation, and premature protecting group loss accounted for over 60% of synthesis failures requiring re-synthesis. Recognizing these problems early saves significant time and resources.
Incomplete Fmoc Deprotection
Symptoms include capped (acetylated) deletion sequences and low UV absorbance at 301 nm. Causes range from peptide aggregation on-resin to insufficient piperidine contact time. Solutions include double deprotection treatments, using chaotropic additives (0.8 M LiCl in DMF), or switching to more aggressive bases like DBU.
Aspartimide Formation
Aspartimide occurs when the side-chain carboxyl of aspartate cyclizes with the backbone nitrogen during base treatment. Sequences containing Asp-Gly, Asp-Ser, or Asp-Asn motifs are especially vulnerable. Backbone amide protection (using Hmb or Dmb groups on the susceptible nitrogen) is the most reliable prevention strategy. Adding 0.1 M HOBt to the piperidine solution also reduces aspartimide formation.
Premature Protecting Group Loss
Trt groups can partially cleave during prolonged synthesis or exposure to mildly acidic conditions. Pbf on arginine occasionally migrates under extended piperidine treatment. Monitoring crude peptide mass spectra for peaks at -242 Da (Trt loss) or -252 Da (Pbf loss) helps identify these issues during quality control.
How Do You Verify Deprotection Quality by HPLC and Mass Spectrometry?
Analytical HPLC and mass spectrometry are the primary tools for confirming complete deprotection. Reversed-phase HPLC on a C18 column can resolve protected from deprotected peptides based on hydrophobicity differences. According to a 2019 best-practices guide in Journal of Peptide Science, LC-MS analysis of crude peptides should achieve baseline resolution between the target mass and major side products for reliable quality assessment.
LC-MS analysis of crude peptides should achieve baseline resolution between the target mass and all major impurities for reliable quality assessment. Retained protecting groups add characteristic mass increments — +56 Da for tBu, +252 Da for Pbf, +242 Da for Trt — enabling rapid identification of incomplete deprotection (Journal of Peptide Science, 2019).
Mass Signatures of Retained Protecting Groups
Each protecting group adds a predictable mass increment. Retained tBu adds +56 Da. Retained Boc adds +100 Da. Retained Trt adds +242 Da. And retained Pbf adds +252 Da. When you see a peak at [M+56] in your mass spectrum, you know you’ve got incomplete tBu removal — most commonly from serine or threonine.
Multiple retained groups produce additive mass shifts. A peptide with one retained Pbf and one retained tBu would show [M+308]. These patterns are diagnostic and help guide re-cleavage or purification decisions.
HPLC Retention Time Shifts
Protected peptides are more hydrophobic than their deprotected counterparts. A retained tBu group typically shifts the HPLC retention time by 1-3 minutes on a standard C18 gradient. Comparing retention times before and after re-cleavage with fresh TFA cocktail confirms whether the additional treatment was effective.
[INTERNAL-LINK: peptide quality documentation → /blog/peptide-research-documentation-standards/]
[INTERNAL-LINK: certificates of analysis → /coas/]
Frequently Asked Questions
Why is Fmoc preferred over Boc in modern peptide synthesis?
Fmoc chemistry uses milder deprotection conditions — 20% piperidine rather than neat trifluoroacetic acid — and avoids the hazardous hydrofluoric acid required for final Boc cleavage. A 2019 Chemical Reviews survey found that roughly 90% of research-scale SPPS now uses Fmoc-based protocols. The base-labile Fmoc group is also easier to monitor via UV absorbance at 301 nm.
Can protecting groups be removed selectively during synthesis?
Yes. Orthogonal protecting groups like Alloc (removed by palladium catalysis) and ivDde (removed by hydrazine) can be cleaved on-resin without disturbing Fmoc or tBu/Boc groups. This selective removal enables on-resin cyclization, branching, and site-specific labeling of complex peptide architectures.
What causes aspartimide formation and how is it prevented?
Aspartimide forms when aspartate’s side-chain carboxyl cyclizes with the backbone amide nitrogen during piperidine treatment. Asp-Gly sequences are the most susceptible. Prevention strategies include backbone amide protection with Hmb or Dmb groups, adding 0.1 M HOBt to the piperidine solution, or using Asp(OtBu)-(Dmb)Gly dipeptide building blocks.
How do you know if global deprotection was complete?
LC-MS analysis reveals retained protecting groups as characteristic mass additions: +56 Da (tBu), +100 Da (Boc), +242 Da (Trt), or +252 Da (Pbf). If these peaks appear, re-treatment with fresh TFA cocktail for an additional 2-4 hours, often with adjusted scavenger composition, typically resolves the issue. Arginine-rich peptides frequently need extended cleavage times for complete Pbf removal.
Conclusion
Protecting group chemistry is the invisible architecture behind every successful Fmoc peptide synthesis. The orthogonal Fmoc/tBu strategy — base for temporary protection, acid for permanent — has remained the dominant approach for over three decades because it works reliably and predictably.
Key points to remember: match side-chain protecting groups to amino acid reactivity, monitor Fmoc removal at 301 nm during every cycle, select the right TFA scavenger cocktail for your sequence composition, and always verify complete deprotection by LC-MS before proceeding to purification. Troubleshooting starts with understanding what each protecting group does and how it fails.
For researchers evaluating peptide quality from suppliers, asking about protecting group strategy and reviewing certificates of analysis for mass spectral data provides meaningful insight into synthesis quality. The difference between a clean synthesis and a problematic one often traces back to protecting group decisions made before the first coupling cycle.
[INTERNAL-LINK: next read — solid-phase vs. liquid-phase synthesis comparison → /blog/solid-phase-vs-liquid-phase-synthesis/]
For research use only. Not for human consumption.
Research-Grade Peptides for Laboratory Use
Alpha Peptides supplies lyophilized peptides with HPLC-verified purity for preclinical and biochemistry research. All compounds are for research use only, not for human consumption.
- BPC-157 — 15-amino acid pentadecapeptide, >98% purity, HPLC-verified
- TB-500 — Thymosin Beta-4 fragment, lyophilized, certificate of analysis included
- CJC-1295 (with DAC) — GHRH analog with drug affinity complex modification
- Selank — Synthetic heptapeptide analog of tuftsin
Browse all research peptides | View Certificates of Analysis