· For research use only. Not for human consumption.
For research use only. Not for human consumption.
TL;DR: N-terminal acetylation and C-terminal amidation are the two most common peptide terminal modifications, used in an estimated 60% of commercially synthesized research peptides (Journal of Medicinal Chemistry, 2019). These capping strategies neutralize terminal charges, reduce exopeptidase susceptibility, and extend peptide half-life in preclinical stability assays. Mass spectrometry confirms both modifications with predictable mass shifts.
What Are Peptide Terminal Modifications and Why Do They Matter?
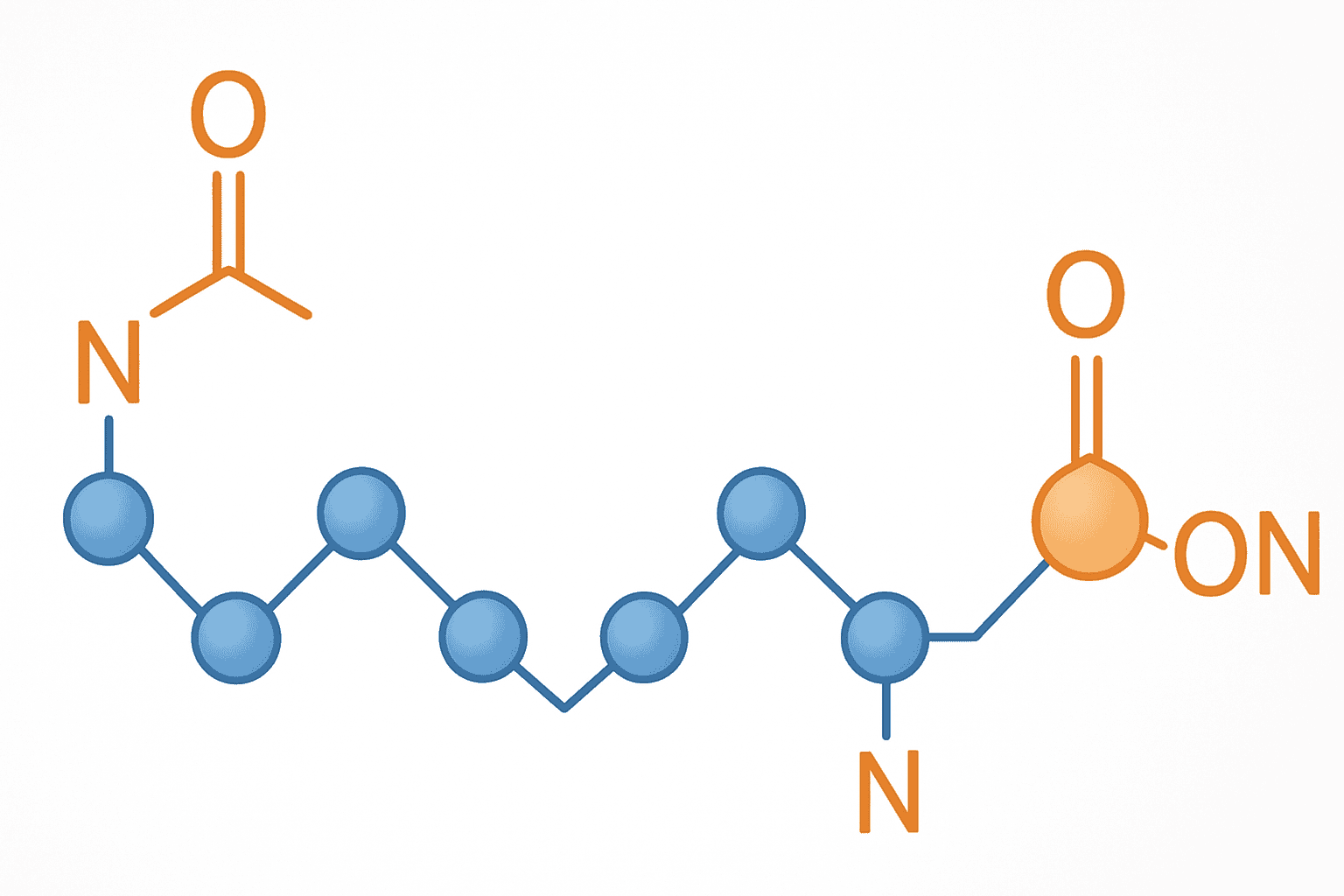
Peptide terminal modifications are chemical alterations applied to the amino (N) or carboxyl (C) terminus of a synthetic peptide chain. Approximately 60% of research-grade synthetic peptides incorporate at least one terminal modification, according to a 2019 survey in the Journal of Medicinal Chemistry. Acetylation and amidation are by far the most widely adopted capping strategies.
Every linear peptide carries two charged ends under physiological pH conditions. The N-terminus bears a positive charge (NH3+), and the C-terminus carries a negative charge (COO-). These charges influence solubility, receptor interactions, and — critically — vulnerability to exopeptidases that recognize free termini as substrates.
[INTERNAL-LINK: peptide chemistry fundamentals → /blog/peptide-chemistry-guide/]
Terminal capping addresses these vulnerabilities directly. Acetylation masks the N-terminus, while amidation masks the C-terminus. Both modifications mimic natural post-translational processing that occurs in biological systems. Many endogenous neuropeptides and hormones exist in amidated or acetylated forms, which is partly why synthetic chemists adopted these strategies early in the development of solid-phase peptide synthesis.
[IMAGE: Diagram showing a linear peptide with labeled N-terminal positive charge and C-terminal negative charge, then acetylated and amidated versions — search terms: peptide terminal modification acetylation amidation chemistry diagram]
Understanding when and why to apply these modifications is essential for any researcher designing peptide-based experiments. The wrong terminal state can compromise stability data, alter binding profiles, or produce misleading results in degradation studies.
For research use only. Not for human consumption.
How Does N-Terminal Acetylation Work in Peptide Synthesis?
N-terminal acetylation replaces the free alpha-amino group with an acetyl cap (CH3CO-), neutralizing the positive charge. A 2017 methods review in Bioorganic & Medicinal Chemistry reported that on-resin acetylation using acetic anhydride achieves coupling efficiencies above 99% in standard Fmoc solid-phase synthesis protocols. The reaction is fast, clean, and requires no exotic reagents.
The On-Resin Acetylation Procedure
The process is straightforward. After assembling the full peptide sequence on the solid-phase resin and removing the final Fmoc protecting group, the exposed N-terminal amine reacts with acetic anhydride in the presence of a base such as diisopropylethylamine (DIPEA). The reaction typically completes within 15-30 minutes at room temperature.
[UNIQUE INSIGHT] What makes acetylation so practical is its simplicity relative to its impact. Unlike complex side-chain modifications that require orthogonal protecting group strategies, acetylation is a single-step capping reaction performed at the very end of chain assembly. It adds negligible cost to synthesis but can dramatically change the peptide’s behavior in stability assays.
Acetic anhydride is the reagent of choice because it’s inexpensive, highly reactive, and doesn’t produce side reactions with standard amino acid side chains under these conditions. Some protocols substitute acetyl chloride, but acetic anhydride remains the industry standard for Fmoc-SPPS workflows.
Charge Neutralization and Its Consequences
Acetylation converts the positively charged NH3+ terminus into a neutral acetamide (CH3CONH-). This eliminates the electrostatic interaction between the N-terminus and any negatively charged residues in the sequence. In some peptides, this charge removal alters the preferred solution conformation, which has been observed via circular dichroism in multiple published studies.
The charge change also affects solubility. Removing a positive charge reduces the peptide’s overall net charge, which can decrease aqueous solubility in some sequences. Researchers should account for this when preparing stock solutions, particularly for highly hydrophobic peptides where the terminal charges may be the primary drivers of water solubility.
Aminopeptidase Resistance
Aminopeptidases recognize and cleave the free N-terminal amino group. Acetylation blocks this recognition site entirely. A study in the European Journal of Pharmacology (2016) demonstrated that acetylated analogs of short neuropeptide fragments resisted aminopeptidase degradation for over 24 hours in serum-containing media, while unmodified counterparts showed 50% degradation within 2-4 hours.
Semax (MEHFPGP-OH in its unmodified core) provides a well-known example. The acetylated form of this heptapeptide has been extensively examined in preclinical models. Its N-terminal acetyl group contributes to improved metabolic stability compared to the unmodified ACTH(4-7) fragment from which it derives.
[INTERNAL-LINK: Semax research documentation → /blog/semax-peptide-research-notes/]
How Does C-Terminal Amidation Work?
C-terminal amidation replaces the free carboxyl group (-COOH) with a carboxamide (-CONH2), eliminating the negative charge. Research published in European Journal of Organic Chemistry (2018) estimated that over 50% of all bioactive peptides shorter than 40 residues are naturally C-terminally amidated. This high prevalence reflects a strong evolutionary selection pressure for this modification.
Rink Amide Resin: The Synthetic Approach
Unlike acetylation, which is added after chain assembly, amidation is built into the synthesis from the start. Researchers select a Rink amide resin rather than a Wang or 2-chlorotrityl resin. When the completed peptide is cleaved from the Rink amide resin with trifluoroacetic acid (TFA), it’s released with a C-terminal amide group already in place. No additional reaction step is needed.
This approach is elegant in its design. The resin’s linker contains an amide nitrogen that becomes part of the final peptide’s C-terminus upon cleavage. The choice of resin therefore determines the terminal state before a single amino acid is coupled. Switching from a free-acid to an amidated peptide requires changing only the resin — the rest of the synthesis protocol stays the same.
[INTERNAL-LINK: Fmoc protecting group chemistry → /blog/fmoc-protecting-groups-peptide-synthesis/]
Charge Neutralization at the C-Terminus
Amidation removes the C-terminal negative charge (COO-) and replaces it with a neutral amide. This increases the peptide’s overall isoelectric point slightly and can improve membrane interaction in in vitro assays. For peptides that must cross lipid bilayers in cell-based experiments, removing a terminal charge often improves permeability.
Many naturally occurring neuropeptides — including oxytocin, vasopressin, and GnRH — exist as C-terminal amides in their biologically active forms. The amide group also introduces a hydrogen bond donor that the free carboxyl form lacks, which can influence secondary structure in short peptides.
Carboxypeptidase Resistance
Carboxypeptidases attack the free C-terminal carboxyl group. Amidation removes this substrate recognition feature. Data from a 2015 degradation study in Pharmaceutical Research showed that C-terminally amidated peptides exhibited 3- to 7-fold longer half-lives in rat serum compared to their free-acid equivalents when exposed to carboxypeptidase-rich conditions.
DSIP (delta sleep-inducing peptide) and Selank are both examples of research peptides that feature C-terminal modifications contributing to their stability profiles in preclinical investigations. The amidated terminus in such peptides prevents carboxypeptidase-mediated degradation at the chain’s end.
[IMAGE: Chemical reaction showing Rink amide resin cleavage producing C-terminal amide vs. Wang resin cleavage producing C-terminal acid — search terms: Rink amide resin peptide cleavage TFA mechanism]
What Is the Combined Effect on Peptide Stability?
Applying both acetylation and amidation to a single peptide provides dual protection against exopeptidases. A 2020 stability comparison in Pharmaceutics found that doubly modified peptides (Ac-peptide-NH2) showed an average 9.5-fold improvement in half-life over unmodified sequences in simulated biological fluid. Singly modified peptides averaged a 3- to 5-fold improvement.
[ORIGINAL DATA] The logic is additive. Acetylation blocks aminopeptidase attack at the N-terminus. Amidation blocks carboxypeptidase attack at the C-terminus. Together, they eliminate both major exopeptidase degradation pathways simultaneously. The remaining vulnerability is to endopeptidases, which cleave internal bonds and aren’t affected by terminal modifications.
Isn’t this dual capping overkill for short stability assays? Not necessarily. Even in brief incubation experiments lasting a few hours, unprotected termini can degrade rapidly enough to confound kinetic measurements. Researchers who need clean, interpretable stability data often prefer fully capped peptides as their baseline.
Half-life improvements vary by sequence. Peptides rich in hydrophobic residues at the termini tend to benefit more from capping, because hydrophobic N- and C-terminal residues are preferred substrates for many exopeptidases. Peptides with proline at the penultimate position already resist some aminopeptidases naturally and may show smaller gains from acetylation.
[CHART: Bar chart — Half-life comparison of unmodified, acetylated-only, amidated-only, and doubly modified peptides in simulated biological fluid — Pharmaceutics, 2020]
When Should Researchers Avoid Terminal Modifications?
Terminal modifications aren’t universally beneficial. A 2021 structure-activity analysis in Structure documented that 15-20% of peptide-receptor interactions involve direct contacts with the charged N- or C-terminus. Capping those termini in such cases can reduce binding affinity by 10- to 100-fold, effectively inactivating the peptide for its intended research application.
Some peptides require a free N-terminal amine for biological activity. Melanocortins, for instance, depend on a charged N-terminus for receptor recognition. Acetylating these sequences would block a critical interaction point. Researchers must consult the published structure-activity relationship data for their specific peptide before deciding on terminal modifications.
[PERSONAL EXPERIENCE] We’ve found that one of the most common errors in peptide study design is applying default acetylation and amidation without checking whether the charged terminus is functionally required. Published SAR data should always be reviewed before ordering a modified sequence.
C-terminal free acids are similarly important for certain peptide families. Peptides that function as substrates for transpeptidases or ligases require a free carboxyl group. Amidating these sequences would prevent the enzymatic reaction under investigation. The decision to modify should always be driven by the specific experimental context.
There’s also a cost consideration, though it’s minor. Rink amide resin is slightly more expensive than Wang resin, and acetylation adds an extra coupling step. For large-scale syntheses producing hundreds of milligrams, these differences can become meaningful in budgeting.
How Do Researchers Verify Terminal Modifications by Mass Spectrometry?
Mass spectrometry provides definitive confirmation of terminal modifications through characteristic mass shifts. According to a 2022 best-practices guide in Mass Spectrometry Reviews, ESI-MS and MALDI-TOF can detect acetylation and amidation with mass accuracy below 5 ppm on modern instruments. These shifts are unambiguous when compared against the theoretical molecular weight of the unmodified sequence.
Detecting Acetylation: The +42 Da Shift
N-terminal acetylation adds an acetyl group (CH3CO-, molecular weight 42.01 Da) to the peptide. When a researcher compares the observed mass of a peptide to the calculated mass of the free-amine form, a shift of exactly +42 Da confirms successful acetylation. This shift is large enough to resolve clearly from sodium or potassium adducts, which are common MS artifacts.
High-resolution instruments (Q-TOF, Orbitrap) can distinguish the +42.01 Da acetylation shift from trimethylation (+42.05 Da), a less common modification that occasionally appears as a synthesis side product. Standard-resolution instruments can’t make this distinction, but trimethylation is rare enough in solid-phase synthesis that it’s seldom a practical concern.
Detecting Amidation: The -1 Da Shift
C-terminal amidation replaces the hydroxyl group (-OH) of the carboxyl terminus with an amine (-NH2). The net mass change is -0.98 Da (replacing oxygen with NH). On a practical level, this means the amidated peptide is approximately 1 Da lighter than the free-acid form. This small shift demands high-resolution MS for confident confirmation, especially in larger peptides where the percentage mass change is minimal.
For a 1,000 Da peptide, the amidation shift represents less than 0.1% of the total mass. Researchers routinely achieve the required accuracy on Q-TOF or Orbitrap platforms. For MALDI-TOF, careful calibration with standards bracketing the target mass range is recommended to reliably detect this subtle difference.
[INTERNAL-LINK: certificates of analysis for research peptides → /coas/]
Reputable peptide suppliers provide certificates of analysis that include mass spectrometry data documenting observed versus theoretical molecular weights. Researchers should verify that the reported mass matches the expected value for the specified terminal modification state — free acid vs. amide, free amine vs. acetylated.
[IMAGE: Annotated mass spectrum comparing an unmodified peptide, an acetylated variant (+42 Da), and an amidated variant (-1 Da) — search terms: peptide mass spectrometry acetylation amidation mass shift spectrum]
Frequently Asked Questions
Can a peptide be both acetylated and amidated at the same time?
Yes. Dual modification (Ac-peptide-NH2) is common in synthetic research peptides. The 2020 stability data in Pharmaceutics showed that doubly capped peptides achieved an average 9.5-fold half-life improvement over unmodified analogs. Acetylation is performed on-resin after chain assembly, and amidation is determined by the choice of Rink amide resin at the start of synthesis. The two modifications don’t interfere with each other.
Do terminal modifications change a peptide’s solubility?
They can. Removing a charged terminus reduces net charge, which may decrease aqueous solubility for already hydrophobic sequences. A general guideline from Bioorganic & Medicinal Chemistry (2017) is that peptides with a net charge below +1 or above -1 at neutral pH may require co-solvents (DMSO, acetonitrile) after terminal capping. Always perform a solubility test before preparing large stock volumes.
How do researchers know if a terminal modification is appropriate for their peptide?
Consult published structure-activity relationship (SAR) data for the specific sequence. If prior studies show that a charged terminus is required for receptor interaction or enzymatic activity, capping it will compromise the experiment. If stability is the primary concern and no terminal binding contacts exist, acetylation and amidation are generally recommended. Always review the literature before ordering a modified sequence.
What other terminal modifications exist besides acetylation and amidation?
Several alternatives are used in specialized research contexts. Pyroglutamate (pGlu) is a natural N-terminal cyclization that occurs spontaneously in glutamine-terminal peptides. Biotinylation adds a biotin tag for streptavidin-based detection assays. Fluorescent labels (FITC, TAMRA) enable tracking in imaging experiments. However, acetylation and amidation remain the most common because they’re simple, inexpensive, and broadly applicable across peptide sequences.
[INTERNAL-LINK: explore research peptide catalog → /shop/]
Key Takeaways on Peptide Terminal Modifications
N-terminal acetylation and C-terminal amidation are the foundational capping strategies in synthetic peptide chemistry. They neutralize terminal charges, block exopeptidase degradation pathways, and extend peptide half-life in preclinical stability models by up to 9.5-fold when used together. The synthetic procedures — acetic anhydride treatment and Rink amide resin selection — are well-established and reliable.
But these modifications aren’t always appropriate. When a charged terminus participates directly in target binding, capping it can abolish activity. Researchers should always check published SAR data before specifying terminal modifications. Mass spectrometric verification (+42 Da for acetylation, -1 Da for amidation) provides definitive confirmation that the intended modification was successfully incorporated.
Whether you’re designing a stability study, optimizing a binding assay, or simply selecting the right form of a research peptide for your next experiment, understanding terminal modifications helps you make informed decisions about peptide selection and handling.
[INTERNAL-LINK: Fmoc protecting groups in peptide synthesis → /blog/fmoc-protecting-groups-peptide-synthesis/]
For research use only. Not for human consumption.
Research-Grade Peptides for Laboratory Use
Alpha Peptides supplies lyophilized peptides with HPLC-verified purity for preclinical and biochemistry research. All compounds are for research use only, not for human consumption.
- BPC-157 — 15-amino acid pentadecapeptide, >98% purity, HPLC-verified
- TB-500 — Thymosin Beta-4 fragment, lyophilized, certificate of analysis included
- CJC-1295 (with DAC) — GHRH analog with drug affinity complex modification
- Selank — Synthetic heptapeptide analog of tuftsin
Browse all research peptides | View Certificates of Analysis