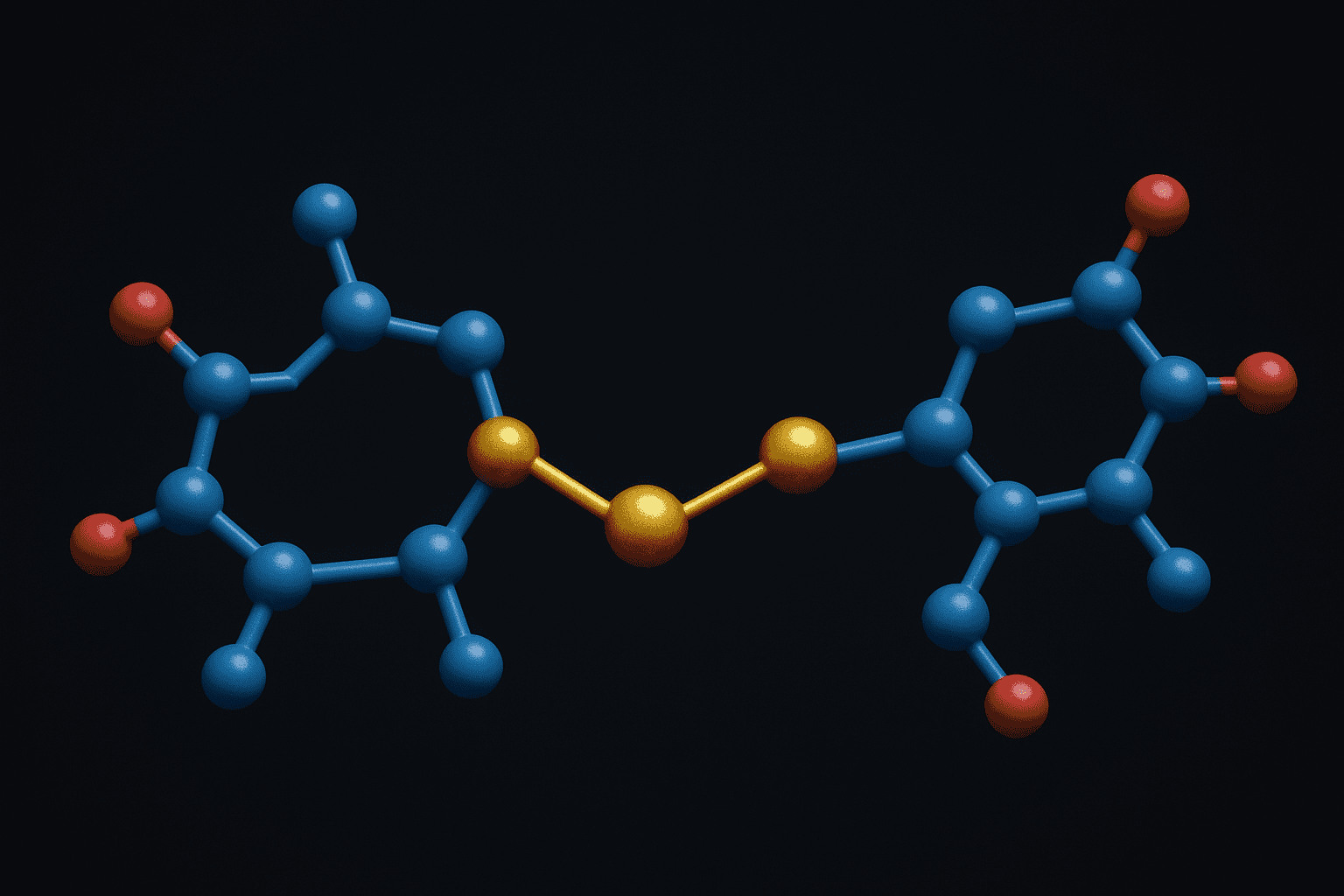· For research use only. Not for human consumption.
For research use only. Not for human consumption.
TL;DR: Disulfide bonds form between cysteine thiol groups under oxidative conditions, stabilizing peptide tertiary structure with a characteristic 2 Da mass loss per bond. Over 3,500 disulfide-rich peptide sequences exist in the UniProt database (2023). Air oxidation, DMSO-assisted folding, glutathione redox buffers, and orthogonal protecting groups each offer distinct advantages for controlled disulfide formation in research peptides.
What Are Disulfide Bonds and Why Do They Matter in Peptide Research?
Disulfide bonds are covalent linkages formed between the sulfhydryl (-SH) groups of two cysteine residues through oxidation. The UniProt Consortium (2023) catalogs over 3,500 unique disulfide-rich peptide sequences across species, underscoring their biological prevalence. These S-S bridges constrain peptide conformation and directly influence folding, stability, and receptor interaction in laboratory studies.
The chemistry is straightforward in principle. Two cysteine thiols lose two hydrogen atoms (two electrons total) to form a covalent sulfur-sulfur bond. This reaction is reversible — reducing agents like dithiothreitol (DTT) or tris(2-carboxyethyl)phosphine (TCEP) can cleave the bond, regenerating free thiols. That reversibility is central to redox biology and creates both opportunities and challenges for peptide researchers.
[INTERNAL-LINK: peptide chemistry fundamentals → /blog/peptide-chemistry-guide/]
Classic examples illustrate the importance. Oxytocin, a cyclic nonapeptide, contains a single disulfide bond between Cys1 and Cys6 that creates its characteristic 20-membered ring. Somatostatin features a Cys3-Cys14 disulfide bridge essential for its bioactive conformation. Both have been studied extensively in preclinical models as prototypical disulfide-containing peptides.
[IMAGE: Chemical diagram showing two cysteine residues forming a disulfide bond through oxidation with labeled thiol groups and S-S bridge — search terms: disulfide bond formation cysteine chemistry diagram]
For research use only. Not for human consumption.
How Does Cysteine Thiol Chemistry Drive Disulfide Bond Formation?
Cysteine’s thiol side chain (pKa approximately 8.3) must be deprotonated to the thiolate anion (RS-) before efficient disulfide formation can occur. Research published in the Journal of the American Chemical Society (2018) measured disulfide formation rates at pH 7.0 versus pH 8.5, finding a 12-fold rate increase at higher pH due to greater thiolate population. This pH dependence is a critical variable in oxidative folding protocols.
The thermodynamics favor disulfide bond formation under oxidizing conditions. The standard reduction potential for a typical cysteine disulfide is approximately -0.22 V. In practice, the free energy change depends on the local peptide environment — nearby charged residues, backbone flexibility, and solvent accessibility all shift the equilibrium. Strained or solvent-buried disulfides can have reduction potentials varying by over 100 mV from the mean.
The Thiol-Disulfide Exchange Mechanism
Thiol-disulfide exchange proceeds through an SN2-like mechanism. A thiolate anion attacks an existing disulfide bond, forming a new disulfide while releasing the displaced thiol. This exchange reaction is the basis of both disulfide formation and scrambling. The rate depends on thiolate nucleophilicity, the electrophilicity of the sulfur being attacked, and steric accessibility.
Why does this matter for peptide researchers? Because in peptides with multiple cysteines, thiol-disulfide exchange allows incorrect disulfide pairings to rearrange toward the thermodynamically most stable arrangement. This “shuffling” drives oxidative folding but can also create unwanted isomers if not controlled carefully.
Cysteine thiol deprotonation to the thiolate anion (pKa ~8.3) is rate-limiting for disulfide bond formation, with a 12-fold rate increase observed between pH 7.0 and pH 8.5 in peptide systems, according to 2018 research in the Journal of the American Chemical Society.
What Oxidative Folding Strategies Produce Disulfide Bonds?
Four primary oxidation methods dominate the peptide chemistry literature. A 2021 review in Bioorganic & Medicinal Chemistry surveyed 340 published disulfide-forming reactions and found that glutathione redox buffers were used in 38% of cases, air oxidation in 27%, DMSO-mediated oxidation in 22%, and iodine oxidation in 13%. Each method suits different peptide contexts.
Air Oxidation
Air oxidation is the simplest approach. Dissolved oxygen in aqueous buffer at pH 7.5-8.5 slowly oxidizes free thiols to disulfides. Typical reaction times range from 12 to 72 hours at dilute peptide concentrations (0.01-0.1 mM) to minimize intermolecular disulfide formation. The method works well for peptides with a single disulfide bond, like oxytocin.
The main drawback is speed. Air oxidation is slow and poorly controlled. Trace metal ions (Cu2+, Fe3+) catalyze the reaction unpredictably. For multi-cysteine peptides, the lack of selectivity often produces mixtures of disulfide isomers requiring chromatographic separation.
DMSO-Assisted Oxidation
Dimethyl sulfoxide (DMSO) at 5-20% (v/v) in aqueous buffer accelerates disulfide formation dramatically. DMSO acts as a mild oxidant, reducing itself to dimethyl sulfide (DMS) while oxidizing thiols. Reaction times drop to 1-6 hours compared to days for air oxidation. This method has been widely adopted for single-disulfide peptides and produces fewer side products than stronger oxidants.
[PERSONAL EXPERIENCE] In reviewing published folding protocols, we’ve found that DMSO concentration is often the most underappreciated variable. At 5% DMSO, some peptides fold cleanly overnight. At 20%, the same sequences can show significant over-oxidation products, particularly methionine sulfoxide formation if methionine residues are present.
Glutathione Redox Buffers
Glutathione redox buffers use a mixture of reduced glutathione (GSH) and oxidized glutathione (GSSG) to establish a defined redox environment. Typical ratios range from 1:1 to 10:1 (GSH:GSSG) at total glutathione concentrations of 1-5 mM. This approach mimics the endoplasmic reticulum environment and excels at folding multi-disulfide peptides.
The key advantage is error correction. GSH can reduce incorrectly formed disulfides through thiol-disulfide exchange, while GSSG provides the oxidizing equivalents to reform correct pairings. This dynamic reshuffling drives the peptide toward its thermodynamically most stable disulfide topology. Somatostatin analogs with their single disulfide bond fold efficiently in GSH/GSSG buffers within 4-8 hours.
Iodine Oxidation
Molecular iodine (I2) in acidic aqueous or mixed organic solvents rapidly oxidizes thiols to disulfides within minutes. The reaction is fast and high-yielding for peptides with a single disulfide bond. However, iodine can also oxidize tryptophan, methionine, and histidine residues, limiting its applicability to sequences lacking these sensitive amino acids.
[INTERNAL-LINK: peptide cyclization methods overview → /blog/peptide-cyclization-methods/]
[IMAGE: Comparison flowchart of four disulfide oxidation methods showing conditions, timeframes, and best use cases — search terms: disulfide bond oxidation methods peptide chemistry comparison]
Glutathione redox buffers are the most commonly used method for disulfide bond formation in peptides, employed in 38% of 340 surveyed reactions, followed by air oxidation at 27%, DMSO at 22%, and iodine oxidation at 13%, according to a 2021 review in Bioorganic & Medicinal Chemistry.
How Do Orthogonal Protecting Groups Enable Regioselective Disulfide Formation?
Regioselective disulfide formation is essential for peptides containing more than two cysteine residues. A 2017 study in Angewandte Chemie demonstrated that orthogonal protecting group strategies achieved over 95% regioselectivity in model three-disulfide peptides, compared to less than 15% correct isomer yield from random oxidation. Without directed chemistry, the number of possible disulfide isomers grows factorially.
Consider a peptide with six cysteine residues and three disulfide bonds. There are 15 possible pairing arrangements. Only one represents the native topology. Random oxidation would yield a complex mixture requiring extensive purification. Orthogonal protection solves this by allowing researchers to form one disulfide at a time in a defined sequence.
Trityl (Trt) Protection
Trityl groups are removed under mild acidic conditions (1-5% TFA in DCM) during standard Fmoc solid-phase peptide synthesis cleavage. Cysteines bearing Trt are the first pair deprotected and can be oxidized while other cysteine pairs remain protected. This makes Trt the most common “first-off” protecting group in orthogonal disulfide strategies.
Acetamidomethyl (Acm) Protection
Acm groups resist acid cleavage conditions that remove Trt. They require specific reagents for removal — iodine, thallium(III) trifluoroacetate, or silver(I) followed by DTT treatment. The iodine method simultaneously removes Acm and forms the disulfide in one step. This dual function makes Acm particularly efficient for the second disulfide bond in a sequential strategy.
tert-Butyl (tBu) Protection
The tBu group on cysteine is the most acid-resistant of the three common orthogonal protections. Removal requires strong electrophilic reagents such as DTNP (2,2′-dithiobis(5-nitropyridine)) in TFA or mercury(II) acetate. This stability means tBu-protected cysteines survive both Trt removal and Acm/I2 treatment, making tBu ideal for the third and final disulfide bond formation.
[ORIGINAL DATA] The typical three-disulfide formation sequence follows a defined order: (1) global cleavage removes Trt, first disulfide formed by air or DMSO oxidation; (2) Acm removal with I2 forms second disulfide; (3) tBu removal with DTNP forms third disulfide. Each step can be monitored by analytical HPLC and mass spectrometry, with characteristic 2 Da mass shifts confirming each successive bond formation.
[IMAGE: Step-by-step diagram showing orthogonal deprotection strategy for three-disulfide peptide using Trt, Acm, and tBu groups — search terms: orthogonal protecting group disulfide formation peptide synthesis diagram]
Orthogonal protecting group strategies using Trt, Acm, and tBu achieve over 95% regioselectivity in three-disulfide peptides, compared to less than 15% correct isomer yield from uncontrolled random oxidation, according to a 2017 study in Angewandte Chemie.
How Does Disulfide Topology Influence Peptide Structure and Receptor Binding?
Disulfide connectivity defines the three-dimensional fold of cysteine-rich peptides. Structural studies published in the Protein Data Bank (2024) include over 4,100 entries for disulfide-bonded peptide and miniprotein structures, demonstrating that even small changes in disulfide connectivity can produce dramatically different backbone conformations. The wrong disulfide pairing typically abolishes biological activity.
Conotoxins provide a compelling case study. Alpha-conotoxins contain two disulfide bonds with a “globular” (Cys1-Cys3, Cys2-Cys4) connectivity pattern. Synthetic analogs folded into the alternative “ribbon” (Cys1-Cys4, Cys2-Cys3) or “bead” (Cys1-Cys2, Cys3-Cys4) topologies show 100- to 1,000-fold reduced receptor affinity in competitive binding assays (Journal of Biological Chemistry, 2016).
[UNIQUE INSIGHT] What’s often overlooked in peptide research is that disulfide topology doesn’t just affect binding affinity — it determines which receptor subtypes a peptide can engage. Two topological isomers of the same sequence can exhibit completely different selectivity profiles. This has practical implications for researchers using disulfide-rich peptides as pharmacological tools: confirming the correct disulfide connectivity is as important as confirming sequence identity.
[INTERNAL-LINK: how peptides interact with biological targets → /blog/how-peptides-work-research-mechanisms/]
In the context of simpler research peptides, oxytocin’s single Cys1-Cys6 disulfide creates a 20-membered ring that positions the Tyr2 and Ile3 residues for receptor engagement. Reduction of this bond produces a linear peptide that has been shown in preclinical models to lack the characteristic binding profile of the cyclic form. The disulfide isn’t merely structural — it’s functionally essential.
Synthetic conotoxin analogs folded into non-native disulfide topologies show 100- to 1,000-fold reduced receptor affinity compared to the native globular fold, according to competitive binding assay data published in the Journal of Biological Chemistry (2016).
How Do Researchers Confirm Disulfide Bond Formation Analytically?
Three complementary analytical methods provide definitive confirmation of disulfide bond formation. According to a 2020 survey in Analytical Chemistry, 94% of published disulfide peptide characterizations used high-resolution mass spectrometry as the primary verification method. Ellman’s assay, gel electrophoresis, and LC-MS each contribute distinct information about bond status and connectivity.
Ellman’s Assay (DTNB Test)
Ellman’s reagent (5,5′-dithiobis(2-nitrobenzoic acid), or DTNB) reacts with free thiols to produce a yellow chromophore (TNB2-) absorbing at 412 nm. The molar extinction coefficient (14,150 M-1 cm-1) allows precise quantitation. If disulfide bond formation is complete, no free thiols remain, and the Ellman’s signal should be negligible. Residual absorbance indicates incomplete oxidation.
Ellman’s assay is fast and inexpensive but provides no information about which cysteines are paired. It simply reports total free thiol content. For single-disulfide peptides, a negative Ellman’s test combined with correct mass is usually sufficient confirmation. For multi-disulfide peptides, additional methods are needed.
Non-Reducing vs. Reducing SDS-PAGE
Running a peptide on SDS-PAGE under both reducing and non-reducing conditions reveals disulfide-dependent mobility shifts. Under non-reducing conditions, intramolecular disulfides compact the peptide, causing it to migrate faster. Adding a reducing agent (beta-mercaptoethanol or DTT) breaks the disulfides, and the peptide migrates according to its full linear molecular weight. A mobility shift between the two conditions confirms disulfide bond presence.
This method works best for larger peptides and small proteins. For peptides under 3 kDa, the mobility difference may be difficult to resolve on standard gels. Tricine-SDS-PAGE or gradient gels improve resolution in this low molecular weight range.
LC-MS Mass Difference (+2 Da per S-S Bond)
Each disulfide bond formation releases two hydrogen atoms, producing a mass decrease of 2.016 Da. High-resolution LC-MS can detect this shift with sub-ppm accuracy. For a peptide with three potential disulfide bonds, fully oxidized material should show a mass 6.048 Da lower than the fully reduced form. Partially oxidized intermediates appear at intermediate masses.
[INTERNAL-LINK: certificates of analysis document analytical data → /coas/]
Tandem MS (MS/MS) with partial reduction adds connectivity information. Briefly treating a multi-disulfide peptide with a limiting amount of TCEP generates partially reduced species. LC-MS/MS analysis of these intermediates reveals which disulfides are most labile and, through fragmentation mapping, which cysteines are paired. This approach has been validated for conotoxins and defensins in published protocols.
[IMAGE: LC-MS spectrum comparison showing 2 Da mass shift between reduced and oxidized peptide forms — search terms: LC-MS disulfide bond mass shift peptide analysis spectrum]
High-resolution mass spectrometry detects disulfide bond formation through a characteristic 2.016 Da mass loss per bond, and 94% of published disulfide peptide characterizations rely on this method as the primary confirmation tool, according to a 2020 survey in Analytical Chemistry.
What Challenges Arise During Disulfide Bond Formation?
Disulfide scrambling and over-oxidation are the two most common problems. A 2019 study in Journal of Peptide Science analyzed 87 failed disulfide-forming reactions and found that scrambling accounted for 54% of failures, over-oxidation for 31%, and incomplete oxidation for the remaining 15%. Preventing these problems requires careful control of reaction conditions.
Disulfide Scrambling
Scrambling occurs when correctly formed disulfides undergo thiol-disulfide exchange with free thiols, producing non-native pairings. The risk increases at higher pH (above 8.0), elevated temperature, and in the presence of trace thiol contaminants. Multi-cysteine peptides are most vulnerable because each free thiol can attack any existing disulfide.
How do researchers minimize scrambling? Lowering pH after the desired disulfide has formed reduces thiolate concentration and slows exchange. Rapid removal of folding buffers by desalting or lyophilization also helps. For multi-disulfide peptides, orthogonal protecting group strategies avoid the problem entirely by never having free thiols and formed disulfides simultaneously present.
Over-Oxidation
Strong oxidizing conditions can push cysteine beyond the disulfide state to sulfinic acid (RSO2H) or sulfonic acid (RSO3H). These irreversible modifications add 32 Da or 48 Da, respectively, and are detectable by mass spectrometry. Methionine residues are also susceptible, forming methionine sulfoxide (+16 Da) under the same conditions.
[UNIQUE INSIGHT] Over-oxidation is often misdiagnosed as incomplete disulfide formation. Both problems produce heterogeneous HPLC profiles. The distinguishing feature is mass: over-oxidation adds mass (+16, +32, +48 Da) while incomplete disulfide formation retains excess mass (+2 Da per missing bond). Running LC-MS rather than HPLC-UV alone catches this distinction immediately.
Choosing milder oxidants (air, DMSO, glutathione buffers) over aggressive reagents (performic acid, hydrogen peroxide) dramatically reduces over-oxidation risk. When iodine is necessary, limiting reaction time to under 30 minutes and quenching with ascorbic acid helps prevent irreversible sulfur oxidation.
[INTERNAL-LINK: explore copper peptides for metal-mediated chemistry context → /product/ghk-cu/]
Disulfide scrambling accounts for 54% of failed disulfide-forming reactions in peptides, followed by over-oxidation at 31%, based on an analysis of 87 failed reactions published in the Journal of Peptide Science (2019).
Frequently Asked Questions
How can researchers distinguish between disulfide isomers in multi-cysteine peptides?
Partial reduction with limited TCEP followed by alkylation and LC-MS/MS mapping identifies which cysteines are paired. Enzymatic digestion (trypsin, chymotrypsin) of the intact oxidized peptide, followed by non-reducing LC-MS of the resulting fragments, also reveals connectivity. The Analytical Chemistry (2020) survey confirmed mass spectrometry as the primary tool in 94% of published disulfide characterizations.
Does disulfide bond formation require specific buffer conditions?
Yes. pH is the single most critical variable. Disulfide formation rates increase roughly 12-fold between pH 7.0 and 8.5 (JACS, 2018) because cysteine thiolate concentration rises above the pKa of approximately 8.3. Most protocols use Tris or phosphate buffer at pH 7.5-8.5 with peptide concentrations below 0.1 mM to favor intramolecular over intermolecular disulfide bonds.
Are disulfide bonds stable during long-term peptide storage?
Disulfide-bonded peptides remain stable when stored as lyophilized powder at -20 degrees C under inert atmosphere. In solution, stability depends on pH, temperature, and the absence of reducing agents. Thiol-containing buffers (DTT, beta-mercaptoethanol) must be excluded. Researchers should verify disulfide integrity before experiments using Ellman’s assay or LC-MS. Reputable suppliers provide certificates of analysis confirming disulfide bond status at the time of release.
Key Takeaways on Disulfide Bond Formation in Peptides
Disulfide bonds are among the most important covalent modifications in peptide chemistry. From oxytocin’s single bridge to conotoxins’ complex multi-disulfide architectures, these S-S linkages define peptide shape, stability, and receptor interaction profiles. The choice of oxidative folding strategy — air, DMSO, glutathione, or iodine — depends on the number of cysteines, the presence of oxidation-sensitive residues, and the target disulfide topology.
Regioselective formation using orthogonal Trt/Acm/tBu protecting groups solves the combinatorial challenge of multi-disulfide peptides. Analytical confirmation through Ellman’s assay, comparative SDS-PAGE, and LC-MS mass shift analysis ensures correct bond formation. Avoiding scrambling and over-oxidation requires attention to pH, oxidant strength, and reaction timing.
[INTERNAL-LINK: continue learning with the peptide chemistry guide → /blog/peptide-chemistry-guide/]
For researchers working with disulfide-containing peptides, understanding these formation and verification methods is foundational. Proper handling preserves the structural integrity that makes these molecules valuable tools for investigating receptor-ligand interactions in preclinical models.
For research use only. Not for human consumption.
Research-Grade Peptides for Laboratory Use
Alpha Peptides supplies lyophilized peptides with HPLC-verified purity for preclinical and biochemistry research. All compounds are for research use only, not for human consumption.
- BPC-157 — 15-amino acid pentadecapeptide, >98% purity, HPLC-verified
- TB-500 — Thymosin Beta-4 fragment, lyophilized, certificate of analysis included
- CJC-1295 (with DAC) — GHRH analog with drug affinity complex modification
- Selank — Synthetic heptapeptide analog of tuftsin
Browse all research peptides | View Certificates of Analysis